East Asian Arch Psychiatry 2021;31:71-80 | https://doi.org/10.12809/eaap2097
ORIGINAL ARTICLE
Ching-Man Lau, Occupational Therapist, Shatin Hospital, Hong Kong
Wai-Kwong Tang, Department of Psychiatry, The Chinese University of Hong Kong, Hong Kong
Address for correspondence: Ching-Man Lau, Occupational Therapy Department, 2/F, Shatin Hospital, 33 A Kung Kok St, Ma On Shan, Hong Kong. Email: 1155121791@link.cuhk.edu.hk
Abstract
Objectives: To investigate the effectiveness of eight 45-minute sessions of computer-assisted cognitive training programme (CCTP) on improving the cognitive and functional performance of patients with Severe Mental Illness (SMI).
Methods: Medical records of 16 women and 13 men aged 26 to 62 (mean, 46.34) years who participated a CCTP were reviewed. The CCTP lasted a total of 6 hours in eight sessions over 8 weeks and comprised a series of mobile applications customised to patients’ specific impaired cognitive domains. Pre- and post- test performance of cognition and functioning were assessed using the Montreal Cognitive Assessment Hong Kong version (HK-MoCA) and the Brief Assessment of Prospective Memory (BAPM), respectively. Results: After the CCTP, the mean HK-MoCA score increased significantly (23.62 ± 5.34 vs 25.48 ± 3.75, d = 0.403, p = 0.001), with a significant increase in delayed recall (3.14 ± 1.75 vs 3.93 ± 1.44, d = 0.493, p = 0.003), and the mean BAPM score decreased significantly (1.44 ± 0.47 vs 1.26 ± 0.23, d = 0.486, p = 0.012). The improvement was greater in participants with primary-level education than in participants with secondary- or tertiary-level education in terms of the HK-MoCA score (3.83 ± 3.06 vs 1.35 ± 2.12, d = 0.942, p = 0.046) and the BAPM scores (-0.49 ± 0.43 vs -0.10 ± 0.29, d = 1.063, p = 0.035).
Conclusion: Our shortened CCTP effectively enhanced the cognitive performance and daily functioning of patients with SMI. Verbal episodic memory showed the most improvement. The improvement was greater in those with primary-level education than in those with secondary- or tertiary-level education.
Key words: Cognitive remediation; Mental disorders; Mobile applications
Introduction
Cognition refers to the mental processes involved in thinking and acquiring information.1 The American Psychiatric Association identifies six key domains of cognitive function: complex attention, executive function, learning and memory, language, perceptual-motor function, and social cognition.2
Cognitive impairment is common in people with severe mental illness (SMI). People with schizophrenia often have cognitive deficits in memory, attention, verbal fluency, visual processing, and psychomotor skills,3 resulting in decreased functioning in daily activities, social participation, and vocational outcomes.4 In people with mood disorders, the impaired cognitive domains include attention, executive function, and memory; greater symptom severity is associated with increased cognitive dysfunction.5
Cognitive rehabilitation for SMI includes pharmacological and behavioural approaches. Although new antipsychotic medications have a more positive effect than traditional medications in improving cognition,6 the efficacy is minimal in terms of functional outcome and community reintegration.7 Behavioural interventions based on the concept of drill and practice have been advocated since 1980s.8 These include mainly strategy-based methods delivered in a verbal or a paper-and-pencil format in group or individual sessions. Improvements in attention, memory, problem-solving, and processing have been observed.
Computer-assisted cognitive training programmes (CCTPs) have advantages of prompt and accurate presentation of stimuli and recording of responses, standardised format, and cost-effectiveness.9,10 CCTPs are effective remedial method for improving executive function, working memory, verbal episodic memory, spatial episodic memory, and processing speed.11 CCTPs can promote neuroplasticity12 (deficit of which may cause social dysfunction13) and foster development of new compensatory skills for tackling cognitive difficulties in daily living.11 After participated in a CCTP, patients with chronic schizophrenia have greater activation in the left frontopolar and left dorsolateral prefrontal cortices, which correspond to improvements in attention and working memory.14 Patients with SMI are reported to have improved functional ability after participating in CCTPs,15 in addition to greater engagement in social rehabilitation programmes and improved employment outcomes.16
Various licensed CCTPs have been developed to target global cognition, speed of processing, attention, working memory, verbal memory, visual memory, reasoning, and problem-solving.17 A 100-hour CCTP with CogRehab for 12 months was reported to improve all cognitive domains, especially working memory.11 Similarly, a 24-hour CCTP with Cogpack was reported to improve verbal memory and processing speed within 12 weeks.18 Whereas 6 hours of training with the Neuropsychological and Educational Approach to Remediation was reported to enhance problem- solving skills.19 Nonetheless, the required licenses pose a barrier to their use. The long training periods may not be applicable to hospital settings in Hong Kong, where the average hospitalisation duration is 7 weeks.20
Some CCTPs have been designed for Hong Kong users such as Six Arts21, Brain Gymnasium,22 and a computerised errorless learning-based memory training programme.10 These CCTPs have positive effects on generalised cognition and prospective memory in daily living but are designed for older patients with dementia and mild cognitive impairment. In adult psychiatric patients, a 12-session CCTP was reported to have no significant effect on cognitive or vocational outcomes.23 This study aimed to investigate the effectiveness of eight 45-minute sessions of CCTP on improving cognitive performance and functional ability of patients with SMI.
Methods
This was a retrospective, single-group, pre-test post-test study. We retrospectively reviewed records of patients with SMI from the Adult Psychiatric Day Hospital in Shatin Hospital who completed the CCTP between May 2018 and May 2019. Inclusion criteria were patients aged ≥18 years with a diagnosis of SMI including schizophrenia (F20), schizoaffective disorder (F25), bipolar affective disorders (F31), and recurrent depressive disorder (F33), according to the ICD-10.24 Exclusion criteria were those with severe auditory or visual impairment, evidence of mental retardation, neurocognitive disorder, traumatic brain injury with a sustained loss of consciousness, or presence or history of epilepsy. Of 31 patients completed the CCTP, two were excluded (one had a history of traumatic brain injury and one had mild neurocognitive disorder) and 29 were included.
Participants’ global cognitive functioning was assessed using the Montreal Cognitive Assessment Hong Kong version (HK-MoCA),25 which covers the cognitive domains of visuospatial ability, executive function, naming, memory, attention, language, abstraction, delayed recall, and orientation. Total score is 30; lower scores indicate greater impairment in cognitive functioning. The MoCA
has good reliability and sufficient concurrent validity in detecting cognitive impairment in patients with SMI. It is an effective bedside screening tool for patients with schizophrenia, psychosis, or mood disorders.26,27
Daily functioning was assessed using the Brief Assessment of Prospective Memory (BAPM).28 This self- report questionnaire is shortened from the Comprehensive Assessment of Prospective Memory. The BAPM has strong agreement with the comprehensive version and good test-retest reliability, internal consistency, and criterion validity.28 The BAPM contains 16 items in two categories: basic activities of daily living (BADL) and instrumental activities of daily living (IADL). Each item is rated in terms of the frequency of forgetting in the last month on a 5-point scale from very often (5) to never (1). Lower scores indicate fewer episodes of forgetting.
Gamified training on personal computers or mobile devices has the advantages of increasing motivation, usability, long-term engagement, ecological validity, and brain stimulation owing to gaming features such as competition, narrative, leaderboards, graphics, and game design.29
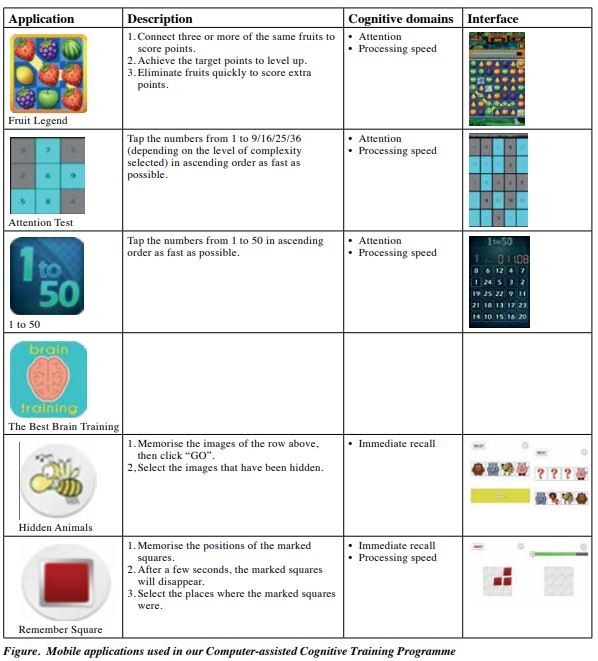
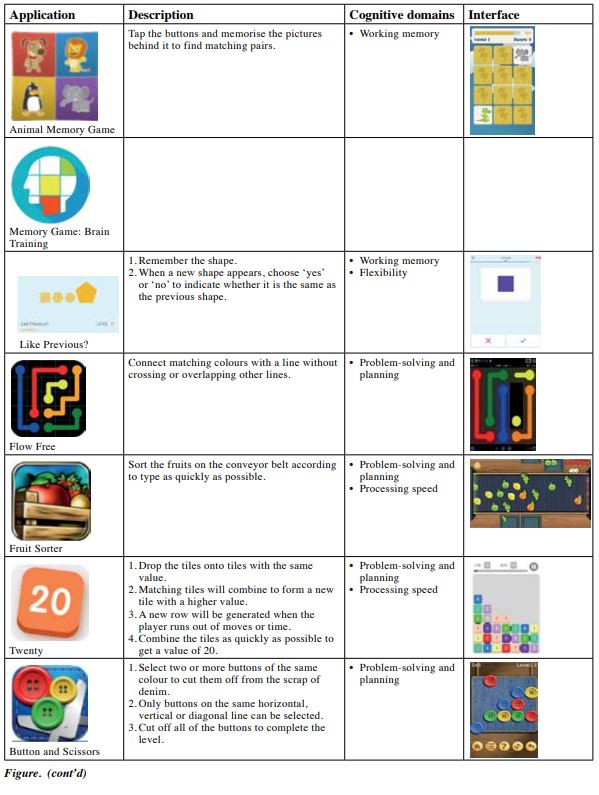
Our CCTP has been used in patients with cognitive defects since 2018. It consists of eight 45-minute sessions and uses mobile applications in tablets. The mobile applications were analysed and categorised by occupational therapists based on the cognitive domains of speed of processing, attention, working memory, verbal memory, visual memory, reasoning, and problem-solving, as suggested in a literature review.17 The activity analyses of all mobile applications are shown in the Figure. Therapists selected the mobile applications according to the deficit areas identified in pre-assessment. Participants were under the guidance and supervision of occupational therapists and trained staff. The training duration and performance were recorded to facilitate monitoring of the treatment progress. The HK-MoCA and BAPM scores before and immediately after the CCTP were compared.
The post-hoc power of the CCTP was computed using AI-Therapy Statistics BETA. The effect size (Cohen’s d)
was calculated according to the equation 
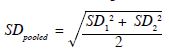
Results
Medical records of 16 women and 13 men aged 26 to 62 (mean, 46.34 ± 12.83) years were reviewed. 16 were young adults or middle-aged adults and 13 were older adults. 16 were diagnosed with schizophrenia, schizotypal, or delusional disorders and 13 were diagnosed with mood disorders. Six had primary-level education and 24 had secondary- or tertiary-level education.
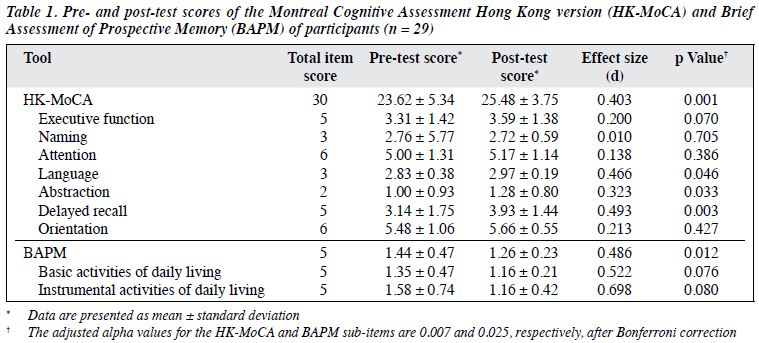
From baseline to post-CCTP, the mean HK-MoCA score increased significantly (23.62 ± 5.34 vs 25.48 ± 3.75, d = 0.403, p = 0.001), with a significant increase in delayed recall (3.14 ± 1.75 vs 3.93 ± 1.44, d = 0.493, p = 0.003), and the mean BAPM score decreased significantly (1.44 ± 0.47 vs 1.26 ± 0.23, d = 0.486, p = 0.012) [Table 1].
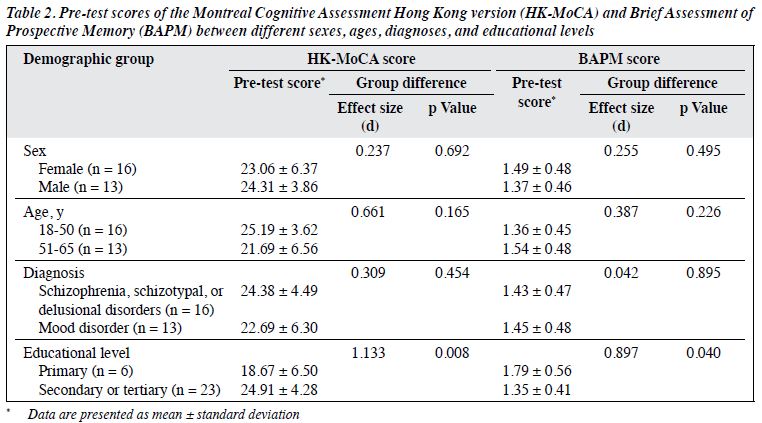
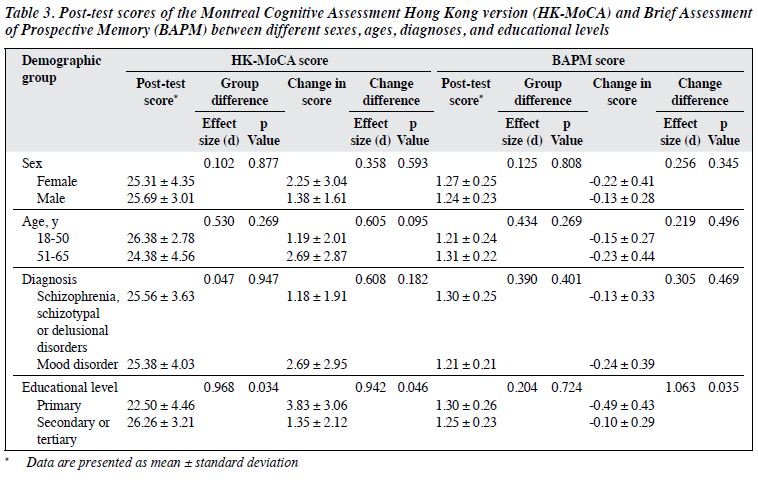
The pre-test HK-MoCA score was significantly better in those with secondary- or tertiary-level education than in those with primary-level education (24.91 ± 4.28 vs 18.67 ± 6.50, d = 1.133, p = 0.008), as was the pre- test BAPM score (1.35 ± 0.41 vs 1.79 ± 0.56, d = 0.897, p = 0.040) [Table 2]. Sex, age, and diagnosis had no significant effect on pre-test HK-MoCA or BAPM score. The post-test HK-MoCA score was significantly higher in those with secondary- or tertiary-level education than in those with primary-level education (26.26 ± 3.21 vs 22.50 ± 4.46, d = 0.968, p = 0.034, Table 3), but post-test BAPM score did not differ significantly between the two subgroups. However, the improvement was greater in those with primary-level education than in those with secondary- or tertiary-level education in terms of the HK-MoCA score (3.83 ± 3.06 vs 1.35 ± 2.12, d = 0.942, p = 0.046) and the BAPM scores (-0.49 ± 0.43 vs -0.10 ± 0.29, d = 1.063, p = 0.035) [Table 3].
Discussion
After the CCTP, cognition, particularly the delayed recall component (involving working memory and verbal episodic memory), and functional performance improved significantly in patients with SMI; these improvements were greater in those with lower educational level. Consistently, other studies reported that 100 hours of CCTP significantly improved working memory and verbal episodic memory among patients with SMI in an outpatient clinic,11 and that 24 hours of CCTP using Cogpack significantly improved overall cognition, learning, and memory in an outpatient setting,16 and that CCTP using Cogpack significantly improved overall cognition and learning in an inpatient setting.18
However, in our study, no significant improvement was found in the other cognitive domains. This contradicts a meta-analysis and other studies that reported significant improvements in abstraction, executive function, attention, and verbal learning after CCTPs.11,12,32 One possible explanation is the small effect sizes of the HK-MoCA sub-scores. For the cognitive components, the meta- analysis obtained a medium effect size range of 0.39 to 0.52,32 whereas the other study had a medium effect size of 0.63.11 In our study, the effect sizes for executive function, naming, attention, language, abstraction, delayed recall, and orientation were 0.20, 0.06, 0.14, 0.48, 0.32, 0.50, and 0.21, respectively. The powers of the sub-scores were 0.436, 0.065, 0.172, 0.533, 0.599, 0.903, and 0.139, respectively. This indicates that the effect sizes of the domains were small to medium, and the corresponding post-hoc powers were modest.
In a meta-analysis of cognitive remediation in schizophrenia, the average duration of cognitive training was 10.4 hours over 12.8 weeks.32 Our CCTP lasted a total of 6 hours over 8 weeks, which is more feasible in Hong Kong where the average hospitalisation is 7 weeks.20 Our shortened CCTP could effectively improve the cognitive and functional performance of patients with SMI. The training content and components were prescribed to patients according to their specific impaired cognitive domains. For studies with longer duration of CCTP, one reported that a 12-hour CCTP improved functional independence among older adults, but specific functional domains were not assessed.33 Another reported that 12-hour CCTP improved attention and resulted in better work outcomes.31 Another reported that a 24-hour CCTP improved performance in competitive employment, including the ability to earn higher wages, increased work tolerance, and the ability to find employment in a broader variety of jobs.16
The improved cognitive performance and functional independence after CCTP may be due to enhanced neuroplasticity. After CCTP, neural imaging shows increases in activation in the left frontopolar and left dorsolateral prefrontal cortices, which is related to attention and working memory.14 The event-related potential components over the occipito-parietal cortex decrease after completion of the CCTP.33 This finding suggests that patients develop an enhanced ability for automatic and spontaneous retrieval in various real-life situations. The effectiveness of cognitive training can extend to everyday tasks. Although neurological assessment tools were not used in our study, the behavioural improvements in cognitive and functioning aspects were evident, which suggests corresponding neurological change.
In our study, the effect of CCTP was similar between patients of different sexes, ages, or diagnoses. However, participants with a lower educational level tended to show a greater improvement in cognitive and functional performance than those with a higher educational level. In other studies, improvement in cognitive performance was greater in younger adults (<40 years) than older adults,34,35 and improved attention in both younger and older adults resulted in better work outcomes.31 However, a meta- analysis reported that older adults gained more than younger adults in terms of cognition and functioning.32 Similarly, older adults were reported to have more room for cognitive improvement.15
Intelligence and educational level are positively related.36 Improvement in cognition and functioning is associated with higher pre-morbid IQ.36,37 In contrast, patients with worse baseline performance have more potential for improvement.15 In the present study, although patients with lower educational level had greater impairment in cognitive and functional performance, they could gain more from the CCTP. Patients with a higher intellectual ability may have a greater cognitive reserve to generalise the skills learnt from the CCTP to reality.37
Nonetheless, the importance of motivation and insight should be emphasised.15 Those who have subjective cognitive complaints and lack compensatory strategies at baseline are more willing to learn and have more potential to improve. Individualised CCTPs focusing on specific cognitive deficits of participants can considerably enhance the related cognitive domains.11 Therefore, the emphasis of CCTPs should be on patients’ drive for training and the customisation of the programme rather than focusing on the individual demographic differences.
This study has some limitations. First, the sample size was small, leading to a wide confidence interval for effect size. The effect sizes were medium (0.403 for HK-MoCA and 0.486 for BAPM) and the statistical powers were insufficient (0.568 for HK-MoCA and 0.759 for BAPM). Second, our patients received multi-disciplinary treatment including medication titration, physiotherapy, vocational rehabilitation, and counselling. Many confounding factors might have affected cognition and functioning, and a control group should have been used. Third, the present study was not blinded, so potential researcher biases were not eliminated. Fourth, the cognitive assessment instrument used might not have been the most appropriate. Although the HK-MoCA is an effective screening tool for SMI,26,27 it does not reveal in-depth and domain-specific neuropsychological information such as processing time and verbal learning, which are commonly impaired in patients with SMI.38,39 Further studies that evaluate long- term effects and include a control group are needed. The use of a greater variety of assessment tools and incorporation of home training elements are also needed. Long-term studies with a control group in various settings are warranted to determine the effectiveness of our CCTP and specifically on symptoms, work outcomes, and social functioning. The levels of motivation and subjective opinions should have been measured. Home training after discharge can be explored, especially most mobile applications are available for free.
Conclusion
Our shortened CCTP effectively enhanced the cognitive performance and daily functioning of patients with SMI. Verbal episodic memory showed the most improvement. The improvement was greater in those with primary-level education than in those with secondary- or tertiary-level education.
Contributors
CML designed the study, acquired the data, analysed the data, and drafted the manuscript. WKT critically revised the manuscript for important intellectual content. CML had full access to the data. All authors contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of interest
All authors have disclosed no conflicts of interest.
Funding/support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability
All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics approval
The study was approved by the Joint Chinese University of Hong Kong-New Territories East Cluster Clinical Research Ethics Committee (Ref: 2019.593). The patients were treated in accordance with the tenets of the Declaration of Helsinki. The patients provided written informed consent for all treatments and procedures.
Acknowledgements
We thank the research assistance team of Prof WK Tang for the technical support and assistance in documentation, occupational therapists and supporting staff from the Shatin Hospital for execution of the programme and collection of data, and participants for their participation.
References
- Wayne W. Psychology Themes and Variations. 9th ed. Wadworth: Cengage Learning; 2013:14.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington: American Psychiatric Association: 2013: 593-5. Crossref
- Green MF, Kern RS, Braff DL, Mintz J. Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the “right stuff”? Schizophr Bull 2000;26:119-36. Crossref
- Tsang HW, Leung AY, Chung RC, Bell M, Cheung WM. Review on vocational predictors: a systematic review of predictors of vocational outcomes among individuals with schizophrenia: an update since 1998. Aust N Z J Psychiatry 2010;44:495-504.
- Trivedi JK. Cognitive deficits in psychiatric disorders: current status. Indian J Psychiatry 2006;48:10-20. Crossref
- Rund BR, Borg NE. Cognitive deficits and cognitive training in schizophrenic patients: a review. Acta Psychiatr Scand 1999;100:85-95. Crossref
- Harvey PD, Green MF, Keefe RS, Velligan DI. Cognitive functioning in schizophrenia: a consensus statement on its role in the definition and evaluation of effective treatments for the illness. J Clin Psychiatry 2004;65:361-72. Crossref
- Spaulding WD, Reed D, Sullivan M, Richardson C, Weiler M. Effects of cognitive treatment in psychiatric rehabilitation. Schizophr Bull 1999;25:657-76. Crossref
- Burda PC, Starkey TW, Dominguez F, Vera V. Computer-assisted cognitive rehabilitation of chronic psychiatric inpatients. Comput Human Behav 1994;10:359-68. Crossref
- Lee GY, Yip CC, Yu EC, Man DW. Evaluation of a computer-assisted errorless learning-based memory training program for patients with early Alzheimer’s disease in Hong Kong: a pilot study. Clin Interv Aging 2013;8:623-33. Crossref
- Kurtz MM, Seltzer JC, Shagan DS, Thime WR, Wexler BE. Computer- assisted cognitive remediation in schizophrenia: what is the active ingredient? Schizophr Res 2007;89:251-60. Crossref
- Hogarty GE, Flesher S, Ulrich R, Carter M, Greenwald D, Pogue- Geile M, et al. Cognitive enhancement therapy for schizophrenia: effects of a 2-year randomized trial on cognition and behavior. Arch Gen Psychiatry 2004;61:866-76. Crossref
- Keshavan MS, Hogarty GE. Brain maturational processes and delayed onset in schizophrenia. Dev Psychopathol 1999;11:525-43. Crossref
- Haut KM, Lim KO, MacDonald A 3rd. Prefrontal cortical changes following cognitive training in patients with chronic schizophrenia: effects of practice, generalization, and specificity. Neuropsychopharmacology 2010;35:1850-9. Crossref
- Twamley EW, Burton CZ, Vella L. Compensatory cognitive training for psychosis: who benefits? Who stays in treatment? Schizophr Bull 2011;37(Suppl. 2):S55-62. Crossref
- McGurk SR, Mueser KT, Pascaris A. Cognitive training and supported employment for persons with severe mental illness: one-year results from a randomized controlled trial. Schizophr Bull 2005;31:898-909.Crossref
- Grynszpan O, Perbal S, Pelissolo A, Fossati P, Jouvent R, Dubal S, et al. Efficacy and specificity of computer-assisted cognitive remediation in schizophrenia: a meta-analytical study. Psychol Med 2011;41:163-73.Crossref
- Lindenmayer JP, McGurk SR, Mueser KT, Khan A, Wance D, Hoffman L, et al. A randomized controlled trial of cognitive remediation among inpatients with persistent mental illness. Psychiatr Serv 2008;59:241-7. Crossref
- Medalia A, Dorn H, Watras-Gans S. Treating problem-solving deficits on an acute care psychiatric inpatient unit. Psychiatry Res 2000;97:79-88.Crossref
- Research Office of Information Services Division, Legislative Council Secretariat of HKSAR. Mental Health Services. 2017. Available at https://www.legco.gov.hk/research-publications/english/1617issh29- mental-health-services-20170626-e.pdf. Accessed 4 October 2019.
- The Hong Kong Alzheimer’s Disease Association. Six Art ® Fun App. Available at https://www.eng.hkada.org.hk/six-arts-fun-apps. Accessed 4 October 2019.
- The Hong Kong Society for the Age. Brain Gymnasium. 2014. Available at https://www.loveyourbrain.org.hk/en/node/319. Accessed 4 October 2019.
- Man DW, Law KM, Chung RC. Cognitive training for Hong Kong Chinese with schizophrenia in vocational rehabilitation. Hong Kong Med J 2012;18(Suppl 6):18-22.
- World Health Organization. International Classification of Diseases for Mortality and Morbidity Statistics (10th Revision). Available at https:// icd.who.int/browse10/2010/en. Accessed 16 July 2021.
- Milani SA, Marsiske M, Cottler LB, Chen X, Striley CW. Optimal cutoffs for the Montreal Cognitive Assessment vary by race and ethnicity. Alzheimers Demen (Amst) 2018;10:773-81. Crossref
- Yang Z, Abdul Rashid NA, Quek YF, Lam M, See YM, Maniam Y, et al. Montreal Cognitive Assessment as a screening instrument for cognitive impairments in schizophrenia. Schizophr Res 2018;199:58-63.Crossref
- Fiorillo A, Carpiniello B, De Giorgi S, La Pia S, Maina G, Sampogna G, et al. Assessment and management of cognitive and psychosocial dysfunctions in patients with major depressive disorder: a clinical review. Front Psychiatry 2018;9:493. Crossref
- Man DW, Fleming J, Hohaus L, Shum D. Development of the Brief Assessment of Prospective Memory (BAPM) for use with traumatic brain injury populations. Neuropsychol Rehabil 2011;21:884-98.Crossref
- Lumsden J, Edwards EA, Lawrence NS, Coyle D, Munafò MR. Gamification of cognitive assessment and cognitive training: a systematic review of applications and efficacy. JMIR Serious Games 2016;4:e11. Crossref
- Cohen J. Statistical Power Analysis for the Behavioral Sciences. 2nd Edition. Taylor Francis; 1988.
- Puig O, Thomas KR, Twamley EW. Age and improved attention predict work attainment in combined compensatory cognitive training and supported employment for people with severe mental illness. J Nerv Ment Dis 2016;204:869-72. Crossref
- McGurk SR, Twamley EW, Sitzer DI, McHugo GJ, Mueser KT. A meta-analysis of cognitive remediation in schizophrenia. Am J Psychiatry 2007;164:1791-802. Crossref
- Rose NS, Rendell PG, Hering A, Kliegel M, Bidelman GM, Craik FI. Cognitive and neural plasticity in older adults’ prospective memory following training with the Virtual Week computer game. Front Hum Neurosci 2015;9:592. Crossref
- Wykes T, Reeder C, Landau S, Matthiasson P, Haworth E, Hutchinson C. Does age matter? Effects of cognitive rehabilitation across the age span. Schizophr Res 2009;113:252-8. Crossref
- Kontis D, Huddy V, Reeder C, Landau S, Wykes T. Effects of age and cognitive reserve on cognitive remediation therapy outcome in patients with schizophrenia. Am J Geriatr Psychiatry 2013;21:218-30.Crossref
- Ritchie SJ, Tucker-Drob EM. How much does education improve intelligence? A meta-analysis. Psychol Sci 2018;29:1358-69. Crossref
- Fiszdon JM, Choi J, Bryson GJ, Bell MD. Impact of intellectual status on response to cognitive task training in patients with schizophrenia. Schizophr Res 2006;87:261-9. Crossref
- Gil-Berrozpe GJ, Sanchez-Torres AM, García de Jalón E, Moreno- Izco L, Fañanás L, Peralta V, et al. Utility of the MoCA for cognitive impairment screening in long-term psychosis patients. Schizophr Res 2020;216:429-34. Crossref
- Coen RF, Robertson DA, Kenny RA, King-Kallimanis BL. Strengths and limitations of the MoCA for assessing cognitive functioning: findings from a large representative sample of Irish older adults. J Geriatr Psychiatry Neurol 2016;29:18-24. Crossref