East Asian Arch Psychiatry 2011;21:115-22
ORIGINAL ARTICLE
严超、刘文华、曹原、陈楚侨
Mr Chao Yan, Neuropsychology and Applied Cognitive Neuroscience Laboratory; Key Laboratory of Mental Health, Institute of Psychology, Chinese Academy of Sciences, Beijing, China.
Dr Wen-Hua Liu, PhD, Faculty of Life Sciences, Sun Yat-Sen University; Faculty of Humanities and Social Sciences, Guangzhou Medical College, Guangzhou, China.
Ms Yuan Cao, Department of Applied Social Studies, City University of Hong Kong, Hong Kong SAR, China.
Dr Raymond C. K. Chan, PhD, Neuropsychology and Applied Cognitive Neuroscience Laboratory, Chinese Academy of Sciences, Beijing, China.
Address for correspondence: Dr Raymond Chan, Institute of Psychology, Chinese Academy of Sciences, 4A Datun Road, Beijing 10010, China.
Tel: (86-10) 6483 6274; Fax: (86-10) 6483 6274; Email: rckchan@psych.ac.cn
Submitted: 3 February 2011; Accepted: 24 May 2011
Abstract
Objectives: In our current research, 2 studies were conducted to investigate self-reported pleasure and approach motivation in individuals with schizotypal personality disorders (SPD) proneness.
Methods: In Study 1, 20 individuals with SPD proneness and 20 non–SPD-prone persons were included in the investigation. In Study 2, 24 SPD-prone and 24 non–SPD-prone individuals took part in our research. In all these individuals, memory and perceptual probabilistic reward tasks, and self-report scales were administered to capture their approach motivation and pleasure experience, respectively.
Results: In both of the 2 studies, individuals with SPD proneness demonstrated more problems with self-reported deficits in pleasure experience than those without SPD proneness. However, there was no difference in approach motivation performance between the groups in Study 1; in Study 2, those with higher levels of anhedonia demonstrated a tendency to even more motivated behaviour.
Conclusions: Approach motivation might be intact in schizotypal-proneness persons, although they had more complaints about their hedonic capacity.
Key words: Motivation; Pleasure; Schizotypal personality disorder
摘要
目的:在这个研究中,我们采用两个实验来探讨分裂型人格障碍倾向群体的自我愉快体验和向动机。
方法:研究一纳入分裂型人格障碍倾向和非分裂型人格障碍倾向各20例;研究二则纳入上述两组群体各24例。记忆和知觉概率化奖励任务,以及自我报告问卷分别用於测量趋向动机和愉快体验。
结果:两项研究显示,分裂型人格倾向的群体较多自我报告愉快体验缺失的问题;趋向动机方面,研究一并未发现两组出现差异,但在研究二中,快感缺失较大的参与者倾向表现更多趋向动机。
结论:儘管分裂型人格倾向群组自我报告较多快感缺失的问题,但他们的趋向动机仍可能是完好的。
关键词:动机、愉快、分裂型人格
Introduction
Schizophrenia is associated with a wide range of emotional and interpersonal impairments, including anhedonia and malfunction in approach motivation.1 Anhedonia is defined as the inability to experience pleasure, whereas approach motivation is defined as the motivation in which the behaviour is driven by positive or desirable events or the possibility of these events happening.2,3 Recent studies suggested that patients with schizophrenia have a deficit in self-reported pleasure experience4-7 and approach motivation.8,9
Recent studies also showed that people with a ‘milder’ non-psychotic forms of the schizophrenia spectrum, such as schizotypal personality disorder (SPD),10,11 share common phenomenological,12 genetic,1,13 and cognitive14 characteristics with patients with schizophrenia. Schizotypal personality disorder is characterised by a need for social isolation, odd behaviour and thinking, and often unconventional belief. The schizotypal personality questionnaire (SPQ) was developed specifically to assess SPD based on the criteria of DSM-III-TR and has high clinical validity and reliability in the general population.15 Inconsistent results were found in the studies of pleasure experiences of SPD. Some studies16-18 reported problems with positive affect among samples with SPD characteristics, while others did not.19 Although most of the existing studies on schizophrenia patients have found an approach motivation deficit,8,9 the number of studies on this issue is still quite limited.20 Therefore, it is necessary to specifically study whether such problems exist in people with SPD proneness.
Anticipatory affective experience and motivated behaviours are closely related.21,22 According to Klein,21 the experience of appetitive pleasure encourages the acting-out of the pursuit for consummatory pleasure. Similarly, according to the Anticipatory Affect Model,22 the anticipation elicited by cues that indicate possible rewards increases positive affect (arousal), which promotes approach behaviour. Moreover, in lab experiments it has been repeatedly found that pleasure experience has a significant effect on approach motivation.23-26 Psychopharmacological studies also suggested that the abnormalities in dopamine system activation found in reward processing in schizophrenia27 may be related to anhedonia.28 Therefore, pleasure experience and approach motivation are taken together in the present research.
The purpose of the current study was to examine the positive emotional experience in SPD in relation to their approach motivation. We adopted a signal-detection reward task involving a differential reinforcement schedule with monetary rewards. Studies using signal-detection paradigms suggested that unequal frequency of rewards between signal and noise stimuli is critical to induce the participant’s approach response to the stimulus with more reward.24,26
The response bias was used in the present study to assess the participant’s approach motivation. The reward paradigm to assess approach motivation has been mainly used in patients with major depression,26,29,30 which showed very good validity and reliability. As far as we know, there is no study using this paradigm based on signal-detection theory in either individuals with schizophrenia or SPD. To address the issues mentioned above, 2 studies were conducted. One was to examine approach motivation based on cognitive function, and the other was to evaluate motivation only based on a perceptual level. The purpose of 2 different paradigms employed was to generally clarify approach motivation in individuals with SPD. We hypothesised that problems might appear in self-reported positive emotional experience in people with SPD proneness. We also hypothesised that people with SPD proneness might display problems in the closely related function of approach motivation.
Study 1
Methods
Participants
Participants were recruited from universities in Beijing and Shanghai through internet advertisements. They were included if they: (1) studied all their major subjects except psychology in university, and (2) had no training or experience with sign language (because our stimuli involved sign language, see Materials below). They were excluded if they had a history or family history of neurological or mental diseases. A total of 81 participants with a mean (standard deviation [SD]) age of 22 (2) years, as well as a mean (SD) duration of education of 15 (2) years, who fulfilled these criteria, were recruited.
Participants were further classified into those with and without SPD proneness using the SPQ.15,31 Participants whose score was in the upper and lower 25th percentiles of score distribution were classified as individuals with and without SPD proneness, respectively. Twenty participants with SPD proneness with a mean (SD) SPQ score of 36 (6) took part in the study. Twenty who were non–SPD-prone with a mean (SD) SPQ score of 13 (4) were screened from all the participants by using the same criteria (bottom 25th percentile). The 2 groups did not differ significantly in terms of age, gender ratio, or education level (Table 1).
Materials
Memory Probabilistic Reward Task
A memory probabilistic reward task adopting a signal- detection model was designed to capture approach motivation. This task based on an asymmetric reward schedule was modified according to the paradigm designed by Pizzagalli et al.26 The asymmetric ratio of rewards (higher frequency of rewards was offered for a correct response for signal than for noise) was used in the reward task because it is considered a good means to induce response bias.24-26 The design of the task consisted of 2 independent variables: magnitude (0 point, 5 points, and 10 points) and reward probability (high and low). For reward probability, high probability of reward was associated with the signal, while low probability was linked to the noise. Meanwhile, accuracy, reaction time, response bias, and discriminating ability were viewed as dependent variables. Response bias reflected one’s decision-making strategy associated with approach motivation, while discriminating ability was used to assess one’s memory processing or the task difficulty.25
Participants were given verbal introductions about the task and were told that the goal was to gain as many points as possible. The reward task consisted of 3 blocks including a no-reward block (0 point), a little-reward block (5 points),
Table 1. Comparison of demographic and clinical assessments between control and schizotypal personality disorder (SPD) proneness groups in Study 1.
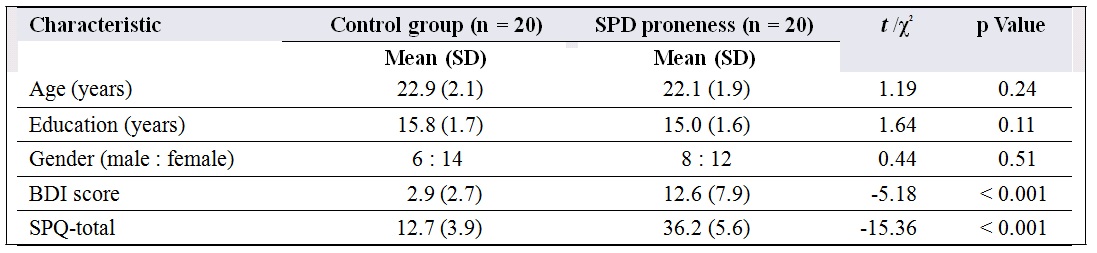
Abbreviations: BDI = Beck Depression Inventory; SPQ-total = total score of Schizotypal Personality Questionnaire; SD = standard deviation.
and a larger-reward block (10 points). Each block was divided into 2 parts: a learning part and a recognition part. Each participant took part in the learning part first, followed by the recognition part. During the learning part, they were told to try their best to memorise 15 pictures which were presented for 2000 ms each. While in the recognition part, there were 90 stimuli (each 300 × 200 px) consisting of both 15 signals which they had learned and 75 pictures (noise) which they had not learned. For each block, all the stimuli were presented in a completely random sequence. Each trial began with a fixation signal at the centre of the screen for 500 ms, and then either a signal or noise would be shown at the centre of the screen for 70 ms. During the 5,000 ms presentation of a mask, participants were asked to identify whether the stimulus had been learned before by pressing the key “J” or the key “F” (Fig 1).
In the reward block, 16 out of 90 trials were followed by reward feedback and these were named reward trials. High reward probability (12 of 16 reward trials) was associated with correct identification of the sign language learned (signal), whereas low reward probability (4 of 16) was linked to correct response of the new sign languages (noise). The reward feedback would be given according to the pseudo-random schedule. If a participant engaged in a reward block (little-reward or larger-reward block) and made a correct identification in a trial for which reward feedback was due according to the schedule, he / she would then receive the feedback of reward. Otherwise, he / she would gain nothing. Before the start of the reward task, it had to be confirmed that each participant understood the rules and knew that not all of correct responses entailed reward feedback but that correct identification of signals would be associated with a higher probability of reward. For each reward block, the feedback was composed of 2 slides: current feedback presented for 2,000 ms (e.g. “Congratulations! You won 5 points!”) and summary feedback for 2,000 ms (e.g. “You have got 25 points in all”). When the task was finished, the points each participant got
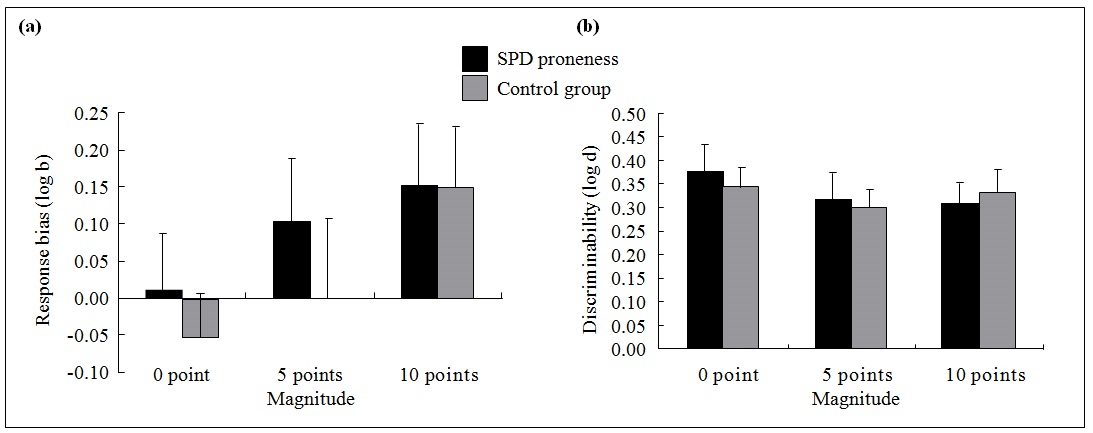
Figure 1. Mean difference in (a) response bias and (b) discriminability between schizotypal personality disorder (SPD) proneness and non–SPD proneness groups in Study 1.*
* Error bars denote standard errors.
would be exchanged for money. For the whole task, each participant would receive RMB20 to 25.
Outcome Measurement
The SPQ,15 a 74-item dichotomous (yes / no) questionnaire was used to screen participants with potential schizotypal reliability (coefficient alpha = 0.91), high test-retest reliability (0.82), and discriminant validity (0.63).15 The Chinese version of the SPQ had been examined in Taiwanese adults and adolescents and found to have good psychometric properties.31
Temporal Experience of Pleasure Scale (TEPS)32 was adopted to assess pleasure experience. It was proved to have good psychometric properties and taped into 2 main factors of pleasure experience: the ‘anticipatory’ factor (e.g. “I get so excited the night before a major holiday I can hardly sleep”) and the ‘consummatory’ factor (e.g. “A hot cup of coffee or tea on a cold morning is very satisfying to me”). The current study adopted the Chinese version of TEPS which demonstrated good clinical discrimination for schizophrenia.33
The Beck Depression Inventory (BDI) was used to measure the severity of symptoms.34 The Chinese version was used for the current study, which proved to have adequate psychometric properties in Chinese samples.35 Anhedonia symptoms were assessed by a subscale in the BDI, including items 4, 12, and 21.36
Statistical Analysis
Trials with reaction times falling outside the range of 3 standard deviations from the mean for each participant were excluded (400 trials, 1.5% of overall trials). Response bias and discriminating ability were computed based on the computation formula illustrated in the research by Pizzagalli et al.26 High value of log b indicated that participants adopted a decision strategy to respond to get more rewards. In other words, a high level of log b suggested that participants were more willing to get a reward and implied high performance of approach motivation. All results were analysed using the Statistical Package for the Social Sciences, Windows version 16.0. Participants with and without SPD proneness were included in the subsequent analysis. In analysing the effect of the SPD on response bias and discriminating ability, two-way repeated measures analysis of variance (ANOVA) was adopted with group as the between-group variable and magnitude as the within- group variable. Significant ANOVA effects of within variables were further analysed using mean comparison of the LSD test. If Mauchly’s test of sphericity was not assumed, the value of Pillai’s trace correction was reported. The value of partial Eta-square (η2) was recorded as effect size. In addition, self-reporting of pleasure experience and the level of depression were analysed using independent sample t tests. The level of statistical significance was chosen as p < 0.05. Effect size on t tests was calculated using Cohen’s d.
Results
Approach Motivation in Individuals with Schizotypal Proneness
For response bias, ANOVA revealed a main effect of personality features. It was found to have high internal magnitude F2,76 = 6.01 (p = 0.004 [partial η
= 0.14]); however, no significant group effect or interaction effect was found (magnitude × group; all p > 0.05). The effect magnitude confirmed the results in the analysis of overall effect in the reward task; further pairwise comparison also showed that response bias for 10 points was higher than that of the 0 point (p < 0.001) [Fig 1a]. For discrimination ability, no significant group effect was shown in the analysis of two-way ANOVA (Fig 1b).
In viewing the group difference of the BDI total scores between SPD-prone and non–SPD-prone groups (t = –5.18, p < 0.001), the total score was considered a co-variable to rule out the effect of depression severity on approach motivation and memory processing. The two-way analysis of co-variance revealed similar results for the analysis of response bias and discriminating ability illustrated above (especially for response bias, no significant group or interaction effect was found; p > 0.05).
Self-reported Pleasure Experience in Schizotypal Proneness
Independent sample t test showed a significant effect of schizotypal features on anhedonia symptoms. The value of Cohen’s d indicated a medium-to-large effect in comparison to the TEPS (anticipatory subscore: Cohen’s d = 0.52; consummatory subscore: Cohen’s d = 0.46; total score: Cohen’s d = 0.52) and the BDI (anhedonia subscore: Cohen’s d = –0.94).
Study 2
The rationale for Study 2 was that the findings in Study 1 might be affected by problems of methodology. First, Study 1 was limited by its small sample size so that the criteria to screen SPD and non–SPD proneness groups were not as strict as in the classification suggested by Raine.15 Second, the task difficulty of the memory probabilistic reward task might have affected the response bias in viewing the main effect of reward magnitude on discriminating ability. Third, no specific self-report scale was adopted to assess anhedonic levels, although the anhedonic subscale of the BDI was used in this study. In view of the findings and limitations of Study 1, we opted to employ a more advanced paradigm to examine approach motivation and self-reported pleasure experience in people with SPD proneness with a more rigorous methodology as described in Study 2.
Methods
Participants
Participants were recruited from universities in Guangzhou
Table 2. Comparison of demographic and clinical assessments between control and schizotypal personality disorder (SPD) proneness groups in Study 2.
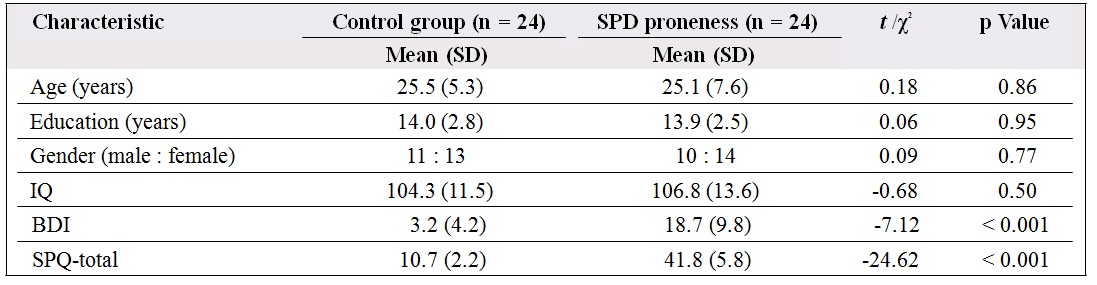
Abbreviations: IQ = The estimate of the Chinese version of the revised Wechsler Adult Intelligence Scale; BDI = Beck Depression Inventory; SPQ-total = total score of Schizotypal Personality Questionnaire; SD = standard deviation.
through internet advertising. A total of 108 participants contacted us and agreed to participate in the study. Following the SPD proneness criterion mentioned in the study of Chan et al,14 24 respondents were identified as schizotypal-prone. The SPD proneness group consisted of 14 women and 10 men with a mean (SD) SPQ score of 41.8 (5.8). A total of 24 participants were selected as non– schizotypal prone persons. The latter group consisted of 13 women and 11 men with a mean (SD) SPQ score of 10.7 (2.2). The 2 groups did not differ significantly in terms of age, gender ratio, education level, or estimated intelligence quotient (Table 2).
Perceptual Probabilistic Reward Task
The perceptual probabilistic reward task was used to measure approach motivation. This task was developed by Bogdan and Pizzagalli,29 and was based on signal-detection theory involving an asymmetric reward schedule. The chief difference was that in the present study the participants were informed of the asymmetric reward rule (as described in Study 1). The reason for this modification was that the current study mainly focused on motivation only, regardless of reward learning. If participants were not informed of the reward rule, they would not only learn the reward rule of the task (reward learning), but also modulate behaviour to the reward stimuli.
The design of the task in Study 2 consisted of 2 independent variables: block (block 1, block 2, block 3) and stimulus type (rich and lean), similar to that in Study Meanwhile, accuracy, reaction time, response bias, and discriminating ability were viewed as dependent variables.
First, the participants were given instructions on a computer monitor. Then they proceeded to the task involving 3 of the same blocks of 80 trials that consisted of 40 rich and 40 lean stimuli. At the beginning of each trial, an asterisk was shown on the computer screen for 1,000 ms, followed by a mouth-less cartoon face. After 500 ms, either a cartoon face with a long mouth (rich stimulus) or a cartoon face with a short mouth (lean stimulus) was displayed for 100 ms. Finally, a mouth-less cartoon face was shown again for 1,500 ms and the participants were asked to identify by pressing the “1”or “4” key, whether a cartoon face with a long mouth or a short mouth had been shown in the previous picture. Throughout the task, an equal number of long and short mouths appeared according to a pseudo-randomised sequence. In addition, reward feedback (e.g. “Congratulations! You got 10 cents!”) was given according to a pseudo-randomised schedule. During each block of trials, a participant received 24 reward feedbacks for the correct identification of the long-mouth stimuli (rich stimuli) but only 8 reward feedbacks for the short-mouth stimuli (lean stimuli).
Participants were informed that not all correct responses were followed by reward feedback. Feedback was not given for a trial if the judgement was wrong or if the judgement was correct but no feedback was scheduled. Only when a response was correct and feedback was scheduled would the participant receive reward feedback. If participants did not respond correctly, they would gain no points even if the trial was scheduled with reward feedback. The reward feedback was presented for 1,500 ms and was followed by a blank screen for 250 ms. If there was no reward feedback, a blank screen was displayed for 1,750 ms (Fig 2a).
To reduce fatigue judgement of the same combination of pictures (mouth), 2 types of cartoon pictures were used (mouth and nose). Thus, there were 2 combinations: long mouth versus short mouth, and long nose versus short nose. The long mouth and short nose were defined as the rich stimuli, and the short mouth and long nose as the lean stimuli. In each block, the first 10 trials presented one combination of cartoon faces (e.g. mouth), and the second 10 trials presented the other combination (e.g. nose), and so on alternately (Fig 2b).
Outcome Measurements
An additional self-report assessment, the Snaith-Hamilton Pleasure Scale (SHAPS)37,38 was adopted to assess the anhedonic state of the participants. The scale included 14 items and assessed an individual’s pleasure experiences in
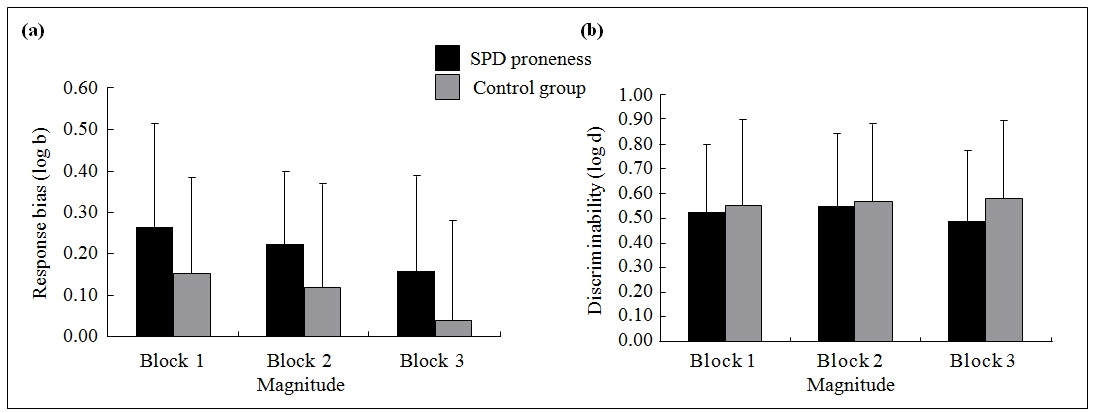
Figure 2. Mean difference in (a) response bias and (b) discriminating ability between schizotypal personality disorder (SPD) proneness and non–SPD proneness groups in Study 2.*
* Error bars denote standard errors.
the last few days. This study used a Chinese translation of the SHAPS with the author’s permission. The Cronbach’s alpha for SHAPS in Chinese samples was 0.85.
The TEPS32,33 was used to evaluate anticipatory and consummatory pleasure experiences among the participants. The Chinese version of the BDI34,35 was also adopted to evaluate the severity of depression and anhedonic symptoms. The SPQ15,31 was used to screen participants with potential schizotypal personality features. Intellectual functions were estimated for all participants by conducting the short form (information, arithmetic, similarity, and digit span) of the Chinese version of the revised Wechsler Adult Intelligence Scale.39 The whole study took participants about an hour, and each obtained RMB 20 to 30 after finishing all the assessments.
Statistical Analysis
Results were analysed using the Statistical Package for the Social Sciences, Windows version 11.5. Independent t tests were used to analyse the differences in age, education level, and intelligence quotient estimates between the 2 groups. The Chi-square test was used to analyse the relationship between gender ratio and schizotypal features. For task and questionnaires, the same statistical methods were adopted as in Study 1.
Results
Approach Motivation in Schizotypal Proneness
Analysis of variance, with block (blocks 1, 2, 3) as the within-group variable and SPD proneness versus non–SPD proneness as the between-group variable, was performed for response bias and discriminating ability, respectively. For response bias, ANOVA revealed a main effect of blocks (F2,45 = 4.7, p = 0.01, partial η = 0.17), with a decreasing response bias from block 1 to block 3 (block 2 < block 1; block 3 < block 1; all p < 0.05). No significant interaction 2 effect emerged (p > 0.05). However, the SPD proneness group seemed to display more response bias than the non– SPD proneness group (p = 0.06; Fig 2). This indicated that the former tended to be more willing to get rewards for the task. For discriminating ability, no significant main or interaction effect emerged (Fig 2).
Furthermore, to rule out the effect of depression severity on response bias, the BDI total score was considered as a co-variable in the analysis. The result was similar. The SPD proneness group displayed comparable response bias as the non–SPD proneness group (p = 0.05).
Self-reported Pleasure Experience in Schizotypal Proneness
For pleasure experience, independent sample t tests were used to test the effect of schizotypal features on anticipatory and consummatory pleasure experiences and on anhedonic levels. Independent sample t tests were also performed to compare the level of hedonic tone for the SPD proneness and non–SPD proneness groups. Result revealed using the SHAPS, the SPD proneness group yielded lower levels of pleasure experience than the non–SPD proneness group (t [46] = –2.87, p = 0.01) and in the BDI-anhedonia subscale (t [46] = –3.97, p < 0.001). There was also a trend of the SPD proneness group to report relatively less anticipatory pleasure than non–SPD proneness group (t [46] = 1.73, p = 0.09). The value of Cohen’s d indicated a medium-to-large effect in the comparisons of TEPS (anticipatory subscore: Cohen’s d = 0.50; consummatory subscore: Cohen’s d = 0.44; total score: Cohen’s d = 0.54), the BDI (anhedonia subscore: Cohen’s d = –1.15), and SHAPS (Cohen’s d = –0.83).
Relationship between Response Bias and the Self-reported Pleasure Experience
Correlation analyses revealed that higher levels of trait anticipatory anhedonia (measured by using the TEPS) were associated with lower levels of response bias in block 1 (r = 0.36, p = 0.08) and block 2 (r = 0.36, p = 0.09). No other significant effect emerged.
Discussion
These 2 studies consistently suggested that individuals with SPD proneness reported higher anhedonic levels, meaning that they experienced relatively less pleasure, especially pleasure in anticipation of future events. This difference approached statistical significance, when compared to those without SPD proneness, which was also consistent with findings in other clinical studies in schizophrenia.28,32 However, no problem was found in the approach motivation of the SPD proneness group, using either the memory or the perceptual signal-detection paradigm, which was inconsistent with research findings in schizophrenics.8,9
The results for pleasure experiences in SPD confirm the compounding findings of psychopathology, theory, and psychometric research. The findings across studies 1 and 2 were consistent with the theoretical postulate suggested by Meehl,1 as well as Watson and Naragon-Gainey.18 They were also compatible with empirical findings in patients with schizophrenia and SPD.17 In previous studies, it was indicated that problems in pleasure experiences including social pleasure were consistently associated with schizotypal personality features and played an important role in the model of schizophrenic spectrum disorder.16-18 Our findings about SPD proneness were also in line with the results of clinical studies in schizophrenia,6,7,40 which indicated that patients with schizophrenia self-reported experiencing less pleasure. In brief, these findings suggest that self-reported pleasure experience may be a promising marker in schizophrenia spectrum disorder.
Notably, the finding that SPD- and non–SPD- prone participants showed similar approach motivation was contrary to our original hypothesis. One possible explanation was that if SPD-prone subjects needed no additional effort to gain the reward, they might show similar approach motivation as the non–SPD-prone (e.g. smoking motivation).41 On the other hand, if they needed more effort to obtain the things they wanted (such as pressing more keys on the keyboard), they might be unwilling to show motivated behaviour. In that sense they might seem to have problems in approach motivation (Chan and Shi, unpublished data), which is also consistent with the clinical studies in schizophrenia.8,9 For the current 2 studies, SPD- prone participants were informed of the asymmetric reward rule, which means that they did not need to take any effort to learn the reward rule. In this case, they showed the same approach motivation as the non–SPD-prone. Thus, it seems that if less effort is needed, approach motivation may remain intact in the SPD-prone persons.
One limitation of the current study was that only a self- report questionnaire was used to assess pleasure experience. Future studies should utilise additional laboratory experiments such as mood induction paradigms to measure pleasure experience in SPD-prone persons objectively. Another limitation is that in the laboratory assessment of motivation, participants made few efforts to make modify responses to the stimulus, because they knew the rules to gain more rewards. In future studies, participants should not be informed of the reward rule in the task so as to assess motivation to apply more effort.
Acknowledgements
This study was supported partially by the Key Laboratory of Mental Health, Institute of Psychology, Chinese Academy of Sciences, the Project-Oriented Hundred Talents Programme (O7CX031003) and the Knowledge Innovation Project of the Chinese Academy of Sciences (KSCX2-YW-R-131 and KSCX2-EW-J-8) from the Institute of Psychology, Chinese Academy of Sciences to Raymond Chan.
References
- Meehl PE. Schizotaxia, schizotypy, schizophrenia. Am Psychol 1962;17:827-38.
- Elliot AJ. Approach and avoidance motivation. In: Elliot AJ, editor. Handbook of approach and avoidance motivation. New York: Psychology Press; 2008: 3-14.
- Elliot AJ, Covington MV. Approach and avoidance motivation. J Educ Psychol 2001;13:73-92.
- Berenbaum H, Oltmanns TF. Emotional experience and expression in schizophrenia and depression. J Abnorm Psychol 1992;101:37-44.
- Blanchard JJ, Mueser KT, Bellack AS. Anhedonia, positive and negative affect, and social functioning in schizophrenia. Schizophr Bull 1998;24:413-24.
- Gard DE, Kring AM, Gard MG, Horan WP, Green MF. Anhedonia in schizophrenia: distinctions between anticipatory and consummatory pleasure. Schizophr Res 2007;93:253-60.
- Horan WP, Kring AM, Blanchard JJ. Anhedonia in schizophrenia: a review of assessment strategies. Schizophr Bull 2006;32:259-73.
- Foussias G, Mann S, Zakzanis KK, van Reekum R, Remington G. Motivational deficits as the central link to functioning in schizophrenia: a pilot study. Schizophr Res 2009;115:333-7.
- Heerey EA, Gold JM. Patients with schizophrenia demonstrate dissociation between affective experience and motivated behavior. J Abnorm Psychol 2007;116:268-78.
- Raine A. Schizotypal personality: Neurodevelopmental and psychosocial trajectories. Annu Rev Clin Psychol 2006;2:291-326.
- 1 Siever LJ, Davis KL. The pathophysiology of schizophrenia disorders: perspectives from the spectrum. Am J Psychiatry 2004;161:398-413.
- Raine A, Reynolds C, Lencz T, Scerbo A, Triphon N, Kim D. Cognitive-perceptual, interpersonal, and disorganized features of schizotypal personality. Schizophr Bull 1994;20:191-201.
- Rado S. Dynamics and classification of disordered behavior. Am J Psychiatry 1953;110:406-16.
- Chan RC, Wang Y, Cheung EF, Cui J, Deng Y, Yuan Y, et al. Sustained attention deficit along the psychosis proneness continuum: a study on the Sustained Attention to Response Task (SART). Cogn Behav Neurol 2009;22:180-5.
- Raine A. The SPQ: a scale for the assessment of schizotypal personality based on DSM-III-R criteria. Schizophr Bull 1991;17:555-64.
- Blanchard JJ, Collins LM, Aghevli M, Leung WW, Cohen AS. Social anhedonia and schizotypy in a community sample: the Maryland longitudinal study of schizotypy. Schizophr Bull 2011;37:587-602.
- Claridge G, McCreery C, Mason O, Bentall R, Boyle G, Slade P, et al. The factor structure of “schizotypal traits”: a large replication study. Br J Clin Psychol 1996;35:103-15.
- Watson D, Naragon-Gainey K. On the specificity of positive emotional dysfunction in psychopathology: evidence from the mood and anxiety disorders and schizophrenia / schizotypy. Clin Psychol Rev 2010;30:839-48.
- Linscott RJ. The latent structure and coincidence of hypohedonia and schizotypy and their validity as indices of psychometric risk for schizophrenia. J Pers Disord 2007;21:225-42.
- Barch DM. Emotion, motivation, and reward processing in schizophrenia spectrum disorders: what we know and where we need to go. Schizophr Bull 2008;34:816-8.
- Klein DF. Depression and anhedonia. In: Clark DC, Fawcett JM, editors. Anhedonia and affect deficit states. New York: PMA Publishing Corporation; 1987: 1-14.
- Knutson B, Greer SM. Anticipatory affect: neural correlates and consequences for choice. Philos Trans R Soc Lond B Biol Sci 2008;363:3771-86.
- Germans MK, Kring AM. Hedonic deficit in anhedonia: support for the role of approach motivation. Pers Individ Dif 2000;28:659-72.
- Henriques JB, Glowacki JM, Davidson RJ. Reward fails to alter response bias in depression. J Abnorm Psychol 1994;103:460-6.
- Miller E, Lewis P. Recognition memory in elderly patients with depression and dementia: a signal detection analysis. J Abnorm Psychol 1977;86:84-6.
- Pizzagalli DA, Jahn AL, O’Shea JP. Toward an objective characterization of an anhedonic phenotype: a signal-detection approach. Biol Psychiatry 2005;57:319-27.
- Simon JJ, Biller A, Walther S, Roesch-Ely D, Stippich C, Weisbrod M, et al. Neural correlates of reward processing in schizophrenia — relationship to apathy and depression. Schizophr Res 2010;118:154-61.
- Dowd EC, Barch DM. Anhedonia and emotional experience in schizophrenia: neural and behavioral indicators. Biol Psychiatry 2010;67:902-11.
- Bogdan R, Pizzagalli DA. Acute stress reduces reward responsiveness: implications for depression. Biol Psychiatry 2006;60:1147-54.
- Pizzagalli DA, Iosifescu D, Hallett LA, Ratner KG, Fava M. Reduced hedonic capacity in major depressive disorder: evidence from a probabilistic reward task. J Psychiatr Res 2008;43:76-87.
- Chen WJ, Hsiao CK, Lin CC. Schizotypy in community samples: the three-factor structure and correlation with sustained attention. J Abnorm Psychol 1997;106:649-54.
- Gard DE, Gard MG, Kring AM, John OP. Anticipatory and consummatory components of the experience of pleasure: a scale development study. J Res Pers 2006;40:1086-102.
- Chan RC, Wang Y, Huang J, Shi Y, Hong X, Ma Z, et al. Anticipatory and consummatory components of the experience of pleasure in schizophrenia: cross-cultural validation and extension. Psychiatry Res 2010;175:181-3.
- Beck AT, Ward CH, Mendelsohn M, Mock J, Erbaugh J. An inventory for measuring depression. Arch Gen Psychiatry 1961;4:561-71.
- Chan CM, Tsoi MM. The BDI and stimulus determinants of cognitive related depression among Chinese college students. Cognit Ther Res 1984;8:501-8.
- Joiner TE, Brown JS, Metalsky GI. A test of the tripartite model’s prediction of anhedonia’s specificity to depression: patients with major depression versus patients with schizophrenia. Psychiatry Res 2003;119:243-50.
- Snaith RP, Hamilton M, Morley S, Humayan A, Hargreaves D, Trigwell P. A scale for the assessment of hedonic tone the Snaith- Hamilton Pleasure Scale. Br J Psychiatry 1995;167:99-103.
- Franken IH, Rassin E, Muris P. The assessment of anhedonia in clinical and non-clinical populations: further validation of the Snaith-Hamilton Pleasure Scale (SHAPS). J Affect Disord 2007;99:83-9.
- Gong YX. Manual of Wechsler Adult Intelligence Scale — Chinese version. Changsha: Chinese Map Press; 1992.
- Burbridge JA, Barch DM. Anhedonia and the experience of emotion in individuals with schizophrenia. J Abnorm Psychol 2007;116:30-42.
- Joseph S, Manafi E, Iakovaki AM, Cooper R. Personality, smoking motivation, and self-efficacy to quit. Pers Individ Dif 2003;34:749-58.


