East Asian Arch Psychiatry 2013;23:154-9
ORIGINAL ARTICLE
谭焕芝、林翠华
Dr Cindy Woon-Chi Tam, FHKAM (Psychiatry), FHKCPsych, Department of Psychiatry, North District Hospital, Hong Kong SAR, China.
Prof Linda Chiu-Wa Lam, FHKAM (Psychiatry), FHKCPsych, MD, Department of Psychiatry, The Chinese University of Hong Kong, Hong Kong SAR, China.
Address for correspondence: Dr Cindy Woon-Chi Tam, Department of Psychiatry, G/F, North District Hospital, 9 Po Kin Road, Sheung Shui, New Territories, Hong Kong SAR, China.
Tel: (852) 2683 7620; Fax: (852) 2683 7616; Email: tamwoonchi@hotmail.com
Submitted: 21 May 2013; Accepted: 9 August 2013
Abstract
Objective: Previous studies have shown that depression is a precursor / prodrome or susceptible state for the development of dementia. This study aimed to examine the relationship between late-onset depression and subsequent cognitive and functional decline in a cohort of non-demented older Chinese persons at their 2-year follow-up and investigate for possible predictors of cognitive decline.
Methods: A total of 81 depressed subjects and 468 non-depressed community controls were recruited. Results: Subjects with late-onset depression showed significantly more incident Clinical Dementia Rating (CDR) scale decline (odds ratio = 3.87, 95% confidence interval = 2.23-6.70) and dementia (odds ratio = 3.44, 95% confidence interval = 1.75-6.77) than those without depression. A higher proportion of depressed CDR 0 subjects had CDR and functional decline than their non-depressed counterparts. Depressed CDR 0.5 subjects had significantly higher rates of functional decline and lower rates of improvement in CDR than their non-depressed counterparts.
Conclusion: Diagnosis of depression was a robust predictor of incident very mild dementia (i.e. CDR of 0.5) and depression severity was a predictor of progression to dementia from CDR of 0.5. The association between depression and the risk of CDR decline and dementia was observed in non-demented Chinese subjects. Depression was also associated with persistent mild cognitive deficits in CDR 0.5 subjects.
Key words: Dementia; Depressive disorder, major
摘要
目的:过往研究显示抑鬱症是老年痴呆症发展的前兆或前驱症状。本文旨在探讨在非老年痴呆症中国老年人群组的2年随访期间,其迟发性抑鬱症与认知和功能衰退的关係,并检视认知能力下降的预测因子。
方法:共纳入81名抑鬱症患者和468名非抑鬱社区老人作对照组。
结果:与对照组比较,迟发性抑鬱症患者较高机会有伴随性临床老年痴呆症评定量表(CDR)表现衰退(比值比 = 3.87,95%置信区间 = 2.23-6.70)和老年痴呆症(比值比 = 3.44,95%置信区间 = 1.75-6.77)。当对照组与两组抑鬱症患者比较,CDR为0的抑鬱症患者有较高比例出现CDR表现衰退和功能下降,而CDR 为0.5的抑鬱症患者则较高机会出现功能下降和较低机会改善CDR。
结论:抑鬱症诊断被认为是伴随性极轻度老年痴呆症(即CDR = 0.5)的强大预测因子,而抑鬱重度则为CDR由0.5进展为1的预测因子。研究指出非老年痴呆症中国老年人其抑鬱症和伴随性CDR表现衰退与老年痴呆症风险的相关性。CDR为 0.5的患者,其抑鬱症与持续性轻度认知障碍也呈相关。
关键词:老年痴呆症、抑鬱症,重性
Introduction
The relationship between cognitive status and depressive symptoms and their effect on functional decline is of clinical and public health significance, because such symptoms and functional disability are common, frequently co-exist, and may be treatable. Previous studies have shown that depression is a precursor / prodrome or susceptible state for the development of dementia.1,2 Depression and the prodrome of dementia are not mutually exclusive, rather they reflect the heterogeneous presentations and aetiologies of both conditions. In the course of depressive disorders, high levels of depressive symptoms are associated with adverse outcomes, including mortality.3
Even in non-demented individuals, both depression and cognitive decline are associated with subsequent onset of dementia. Individuals with mild cognitive impairment (MCI) are at an increased risk of developing dementia. According to a systematic review, the mean conversion rate from MCI to dementia was 10.24% which is 5 times the expected rate in persons of any age.4 Depression in conjunction with MCI has been particularly implicated as a prognostic indicator for Alzheimer’s disease (AD). Depression co-existing with cognitive impairment or dementia has an addictive effect on adverse outcomes for physical health, functional status, and mortality.5 In a community general hospital, Modrego and Ferrández6 found that after a mean period of 3 years, patients with MCI and a clinical diagnosis of depressive disorder were at more than twice the risk of developing AD as those without depression.
In a 12-year longitudinal study, Ganguli et al7 reported that depressive symptoms were cross-sectionally associated with cognitive impairment but not subsequent cognitive decline. Substantial cognitive decline over time cannot be explained by depression alone and most likely reflects incipient dementia. Research has yielded mixed findings of the association between depression and cognitive decline and dementia. Such inconsistent results could have arisen from varying definitions of depression or depressive symptomatology, dementia, and cognitive decline, as well as differences in sample size and durations of follow-up.
In Hong Kong, studies of co-morbid depression and syndromal cognitive impairment are limited, as studies on depression may have excluded or inadequately assessed cognitively impaired participants, and studies of MCI may also have excluded or inadequately assessed syndromal depression. Therefore, the study aimed to establish the rate of incident cognitive decline and dementia in non-demented elderly persons with late-onset depression and to identify the predictors of cognitive decline.
Methods
Depressed Subjects
Chinese patients aged ≥ 60 years who fulfilled the DSM-IV- TR criteria8 for major or minor depression were recruited from a regional psychiatric service in Hong Kong. They had the onset of first depressive episode at the age of ≥ 50 years.
Each depressed subject was evaluated by a qualified psychiatrist to establish eligibility for inclusion in the study, a clinical diagnosis, and an assessment of clinical staging using the Clinical Dementia Rating (CDR) scale.9 Subjects with a global CDR score of 0 or 0.5 were recruited.
In all, a convenience sample of 120 patients with depression were screened from January 2007 to December 2009, 109 fulfilled the recruitment criteria of late-onset depression, but 3 (3%) declined to participate. The remaining 106 subjects completed the first assessment and 1 dropped out thereafter as the subject moved to an old-age home in another catchment area. Thus, 105 subjects had an assessment at 24 weeks, whilst 81 (77%) completed the assessment at 2 years, 3 (3%) had died of physical illness, and 21 (20%) did not have the 2-year assessment because they defaulted follow-up, moved into another catchment area, or refused to be assessed. There were no significant differences between the dropouts and those who completed the 2-year assessment with respect to baseline age, gender, and education level.
Controls
Control subjects were recruited from a population- based epidemiology study of cognitive impairment in the elderly conducted from October 2005 to July 2006.10 All of them were aged ≥ 60 years and had been assessed by an experienced psychiatrist. They were not clinically demented or depressed at baseline nor did they have a history of depression. Control subjects with a global CDR score of 0 or 0.5 were recruited, 468 of whom were age- and education-matched with the depressed subjects and completed the 2-year assessment.
Other Subject-related Aspects
Subjects with a history of degenerative neurological disorder, dementia, cortical strokes, severe or unstable physical illness, and any current or prior substance / alcohol abuse were excluded. Those who had received electroconvulsive therapy in the past 3 months were also excluded. The psychiatrists explained the procedure and obtained informed written consent from the participants or their caregivers. This study was approved by the Joint Clinical Research Ethics Committee of The Chinese University of Hong Kong and the New Territories East Cluster hospitals.
Assessments
All subjects underwent a comprehensive psychiatric, cognitive, and functional assessment. Global cognitive ability was measured by the Mini-Mental State Examination (MMSE)11 and Alzheimer’s Disease Assessment Scale Cognitive Subscale (ADAS-Cog).12 Episodic memory was measured by the 10-minute delayed recall of the word list in the ADAS-Cog. Executive tests, including category verbal fluency test (CVFT)13 and similarity and difference tests, were performed. Working memory was measured by digit and visual span tests. Depression was diagnosed according to the DSM-IV-TR criteria8 and symptom severity was rated using the Neuropsychiatric Inventory (NPI)14 depression subscale. Functional ability was measured based on the Chinese version of the Disability Assessment for Dementia (DAD) instrument.15
The medical illness burden was assessed by Cumulative Illness Rating Scale (CIRS).16 The CIRS is a measure of physical illness in which a cumulative score is derived from ratings of impairment severity in each of 13 organs / systems. In addition, presence of cardiovascular risk factors and vascular diseases including hypertension, diabetes mellitus, hyperlipidaemia, heart disease, arrhythmia, and cerebrovascular accidents were recorded. A combined cardiovascular risk factor (CVRF) score was calculated by adding the total number of risk factors.
The follow-up assessments were the same as at baseline assessment and conducted 2 years later. Dementia was diagnosed according to DSM-IV-TR criteria.8 The CDR 0.5 subjects at baseline were categorised into 3 groups (improved, stable, and declined) based on how their 2-year CDR scores had changed. Subjects with a CDR of 0 at baseline were therefore categorised into stable and declined groups.
Independent variables including age, education, baseline cognitive test scores (MMSE, ADAS-Cog, delayed recall, CVFT, digit span, visual span), functional assessment (DAD), mood (NPI depression) scores, CVRF scores, CIRS scores, and duration of depression were entered into the regression analysis. This was to look for predictors of incident dementia and CDR decline. Significant declines in MMSE and DAD scores at the 2-year follow-up were defined as drops in the respective score of > 1 standard deviation of the baseline mean score from the control with a CDR score of 0.
Statistical Analyses
In the univariate analyses, baseline characteristics of subjects were compared using Mann-Whitney U tests for continuous variables and Chi-square tests for categorical variables. Rates of incident CDR decline and dementia in the depressed and control groups were compared. Mann- Whitney tests were performed to compare the demographic data, clinical characteristics, including medical and vascular risk factor burdens, cognitive test scores, and DAD scores in those who had incident CDR decline or dementia and those who did not. Variables that yielded statistically significant differences were entered as independent variables in the regression analyses. Logistic regression analyses were performed for dichotomous dependent variables (incident CDR decline and dementia). The level of statistical significance was set at p < 0.05 using a 2-tailed distribution. All analyses were performed using the Statistical Package for the Social Sciences Windows version 17.0 (SPSS Inc., Chicago [IL], US).
Results
The baseline characteristics of the depressed subjects and controls are shown in Table 1. The 2 groups were matched in age and educational status, and there were no significant differences between them with respect to ratios of CDR scores of 0 and 0.5, and baseline medical and vascular risk factor burdens. The depressed group had lower MMSE and DAD scores at baseline.
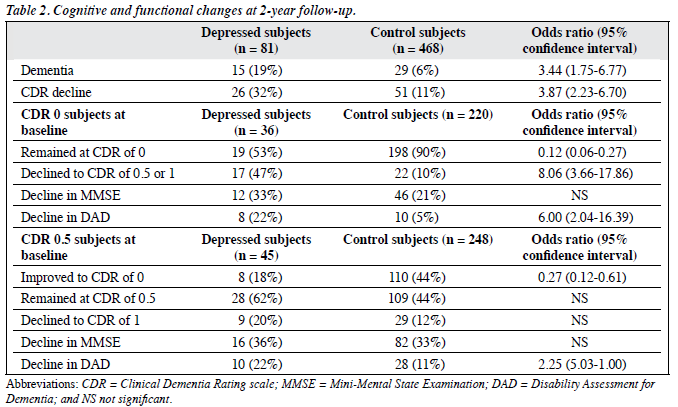
Table 2 shows the cognitive and functional changes in both the depressed and control subjects at 2 years. At 2 years, there was an overall rate of conversion to dementia in 19% of the depressed elderly and 6% in the non-depressed controls; respective odds ratio was 3.87 for CDR score decline and 3.44 for dementia when comparing depressed subjects with the controls. Among subjects with CDR scores of 0 at baseline, none of the controls and 6 (17%) of the depressed subjects progressed to dementia at the 2-year follow-up. Depressed subjects with baseline CDR scores of 0 had significantly higher odds of CDR and functional decline than their non-depressed counterparts. Depressed subjects with CDR scores of 0.5 had a significantly higher odds for functional decline and a significantly lower odds for improvement in CDR scores than their non-depressed counterparts.
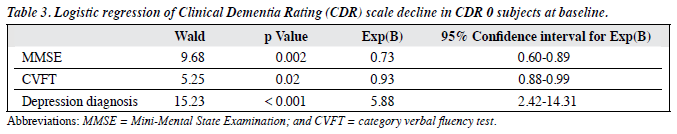
According to the regression analyses, a diagnosis of depression, as well as lower baseline CVFT and MMSE scores predicted a higher risk of CDR decline in subjects with baseline CDR scores of 0. Among subjects with baseline CDR scores of 0.5, depression severity rather than the diagnosis of depression predicted a higher risk of dementia progression. Age and lower baseline CVFT scores also predicted a higher risk or progression from a CDR of 0.5 to clinical dementia.
Discussion
The conversion rate to dementia after 2 years among our CDR 0.5 subjects (19% for depressed subjects and 6% for non-depressed controls) was similar to those reported in other studies. Daly et al17 found an annual conversion rate of 6.3% in CDR 0.5 subjects, whilst Grober et al18 estimated a 6.2% MCI annual conversion rate and Ravaglia et al19 reported it to be 29% over a 3-year follow-up.
Late-onset depression was associated with increased risk of progression to very mild dementia and clinical dementia in 2 years among elderly with a CDR score of 0 at baseline. The association between depressive symptoms and incident MCI has also been reported in the literature.20-22 In our study, the risk of CDR decline was about 8 times higher than those who were not depressed. The risk of CDR decline was also associated with a higher risk of functional decline without any significant decrease in MMSE performance. In subjects with CDR scores of 0.5, a diagnosis of depression was associated with higher risk of functional decline. Baseline elevated depressive symptoms also appeared to predict increased disability in several other longitudinal community studies.23,24 The present findings supported the notion that late-life depression had detrimental effects on the long-term functional ability.
While there was no significant difference in the conversion rate from a baseline CDR score of 0.5 to dementia between the depressed and non-depressed groups, a significantly higher proportion of non-depressed subjects converted back to CDR score of 0. This suggested that depressed subjects tended to have persistent cognitive impairment, though they might not progress to dementia within 2 years. Clinical and population-based studies have shown that MCI in depression often persists after remission of depression. Köhler et al25 found that half of the patients exhibited a generalised cognitive impairment that persisted after 18 months. Four-year follow-up data suggested that impairments persisted but did not decline further. Bhalla et al26 found that 45% of late-life depression subjects were cognitively impaired, despite remission of depression, whilst 94% of the patients who were impaired at baseline remained impaired 1 year later.
After taking into account of covariates (age, education level, baseline cognitive and functional scores), diagnosis of depression or depressive severity and executive dysfunction were found to predict decline in subjects with baseline CDR scores of 0 and 0.5. The influence of depression was more prominent in the elderly with CDR scores of 0. Gender, education, medical burden, and vascular risk factors were not significant predictors. Previous studies also supported our findings of no association between socio-demographic variables or vascular risk factors and conversion to dementia.21,27
The severity of rather than the diagnosis of depression was an independent predictor of incident dementia among subjects with baseline CDR scores of 0.5. We speculate that some community controls had subsyndromal depressive symptoms which also increased risk of progression to dementia from a CDR of 0.5, as reported in the literature. Lyness et al28 found that subsyndromal forms of depression in older adults were associated with significant functional impairment. Rosenberg et al29 also reported subsyndromal depressive syndrome to be a risk factor for the development of cognitive impairment in a longitudinal study of older community-dwelling women. Other research indicates that co-existence of depression and MCI appears to increase the risk for persistent cognitive impairment and dementia.30,31 Taken together, these findings support a trait effect on neurocognition, most probably caused by structural cerebral changes. Such findings have been consistently reported in depression, especially the late- onset type.32,33
The community-defined cohort of the Italian Longitudinal Study on Aging did not find a significant association between depressive symptoms and incident MCI.27 The same group also found that depressive symptoms failed to predict conversion of MCI to dementia.34 Rozzini et al35 also reported that concurrent depression did not appear to increase the risk of AD over 2 years (when compared with non-depressed MCI patients). These contrasting findings suggest that the duration of follow-up, the study design, the population sampled, the level of subject education, the setting, and methodological difference (measurement of depressive symptoms or diagnosis of depressive disorders and level of depressive symptoms) may be crucial. Similarly, inclusion of MCI patients in samples might be the source of conflicting results. Depressive symptoms detected in epidemiological studies of the population at large may represent a milder and less homogeneous affect state than patients with major depression who seek treatment in psychiatric facilities.
This was the first study to examine the association between late-onset depressive syndrome and conversion to dementia in Chinese older persons in Hong Kong. Its strength was the inclusion of both a clinic sample with late- onset depressive syndromes as well as a community sample with subsyndromal depressive symptoms. Moreover, subjects with and without MCI were separately analysed to provide a more comprehensive picture of the contribution of depression and MCI in predicting dementia.
One limitation of the current study was the small sample size of the depressed subjects, especially after the division into CDR of 0 and 0.5 subgroups. Moreover, there was a significant dropout rate at the 2-year follow- up assessment. Dropout subjects might have depressive moods and / or cognitive impairment and lack motivation to participate in the assessment. Hence, our results might underestimate the liability to cognitive decline. Moreover, this was a prospective study with a naturalistic treatment approach. The effects of antidepressants on the cognitive test scores or outcomes were not assessed due to the small sample size and great variety of treatments for depressed subjects. We did not exclude the controls who had incident depression or antidepressant treatment during the 2-year follow-up, but the number was very small compared with the entire sample size. Furthermore, the psychiatrist who carried out the 2-year assessments was not blinded to the diagnosis of depression. The diagnosis of dementia was a clinical diagnosis made by the psychiatrist. To reduce bias, the standardised and objective cognitive and functional assessments were nevertheless performed by a research assistant who did not know the subjects’ diagnosis. We did not exclude the subjects who were taking anti-dementia medications at the time of the 2-year assessment, but the psychiatrist took account of such a clinical history and the medications taken when making the diagnosis of dementia.
Conclusion
We conclude that late-onset depression was associated with a higher risk of CDR score and functional decline over a 2-year period. This study found that depression and executive dysfunction are independent predictors of incident CDR decline and dementia. In addition, late-onset depressive syndrome is a more robust predictor of incident functional and CDR decline in Chinese elderly with baseline CDR scores of 0 than that of 0.5.
Acknowledgement
The authors declared no conflict of interest in this study.
References
- Devanand DP, Sano M, Tang MX, Taylor S, Gurland BJ, Wilder D, et al. Depressed mood and the incidence of Alzheimer’s disease in the elderly living in the community. Arch Gen Psychiatry 1996;53:175-82.
- Green RC, Cupples LA, Kurtz A, Auerbach S, Go R, Sadovnick D, et al. Depression as a risk factor for Alzheimer disease: the MIRAGE study. Arch Neurol 2003;60:753-9.
- Ganguli M, Dodge H, Mulsant BH. Rates and predictors of mortality in an aging, rural, community-based cohort: the role of depression. Arch Gen Psychiatry 2002;59:1064-52.
- Bruscoli M, Loverstone S. Is MCI really just early dementia? A systematic review of conversion studies. Int Psychogeriatr 2004;16:129-40.
- Mehta KM, Yaffe K, Langa KM, Sands L, Whooley MA, Covinsky KE. Additive effects of cognitive function and depressive symptoms on mortality in elderly community-living adults. J Gerontol A Biol Sci Med Sci 2003;58:M461-7.
- Modrego PJ, Ferrández J. Depression in patients with mild cognitive impairment increases the risk of developing dementia of the Alzheimer type: a prospective cohort study. Arch Neurol 2004;61:1290-3.
- Ganguli M, Du Y, Dodge HH, Ratcliff GG, Chang CC. Depressive symptoms and cognitive decline in late life: a prospective epidemiological study. Arch Gen Psychiatry 2006;63:153-60.
- American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders, text revision. 4th ed. Washington, DC: American Psychiatric Association; 2000.
- Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL. A new clinical scale for the staging of dementia. Br J Psychiatry 1982;140:556-72.
- Lam LC, Tam CW, Lui VW, Chan WC, Chan SS, Wong S, et al. Prevalence of very mild and mild dementia in community-dwelling older Chinese people in Hong Kong. Int Psychogeriatr 2008;20:135-48.
- Chiu HF, Lee HC, Chung WS, Kwong PK. Reliability and validity of the Cantonese version of the Mini-Mental State Examination — A preliminary study. Journal of Hong Kong College of Psychiatrists 1994;2:25-8.
- Chu LW, Chiu KC, Hui SL, Yu GK, Tsui WJ, Lee PW. The reliability and validity of the Alzheimer’s Disease Assessment Scale Cognitive Subscale (ADAS-Cog) among the elderly Chinese in Hong Kong. Ann Acad Med Singapore 2000;29:474-85.
- Lam LC, Ho P, Lui VW, Tam CW. Reduced semantic fluency as an additional screening tool for subjects with questionable dementia. Dement Geriatr Cogn Disord 2006;22:159-64.
- Leung VP, Lam LC, Chiu HF, Cummings JL, Chen QL. Validation study of the Chinese version of the neuropsychiatric inventory (CNPI). Int J Geriatr Psychiatry 2001;16:789-93.
- Mok CC, Siu AM, Chan WC, Yeung KM, Pan PC, Li SW. Functional disabilities profile of Chinese elderly people with Alzheimer’s disease — A validation study on the Chinese version of the disability assessment for dementia. Dement Geriatr Cogn Disord 2005;20:112-9.
- Conwell Y, Forbes NT, Cox C, Caine ED. Validation of a measure of physical illness burden at autopsy, the Cumulative Illness Rating Scale. J Am Geriatr Soc 1993;41:38-41.
- Daly E, Zaitchik D, Copeland M, Schmahmann J, Gunther J, Albert M. Predicting conversion to Alzheimer disease using standardized clinical information. Arch Neurol 2000;57:675-80.
- Grober E, Lipton RB, Hall C, Crystal H. Memory impairment on free and cued selective reminding predicts dementia. Neurology 2000;54:827-32.
- Ravaglia G, Forti P, Maioli F, Martelli M, Servadei L, Brunetti N, et al. Conversion of mild cognitive impairment to dementia: predictive role of mild cognitive impairment subtypes and vascular risk factors. Dement Geriatr Cogn Disord 2006;21:51-8.
- Lopez OL, Jagust WJ, Dulberg C, Becker JT, DeKosky ST, Fitzpatrick A, et al. Risk factors for mild cognitive impairment in the Cardiovascular Health Study Cognition Study: part 2. Arch Neurol 2003;60:1394-9.
- Barnes DE, Alexopoulos GS, Lopez OL, Williamson JD, Yaffe K. Depressive symptoms, vascular disease, and mild cognitive impairment: findings from the Cardiovascular Health Study. Arch Gen Psychiatry 2006;63:273-9.
- Geda YE, Knopman DS, Mrazek DA, Jicha GA, Smith GE, Negash S, et al. Depression, apolipoprotein E genotype, and the incidence of mild cognitive impairment: a prospective cohort study. Arch Neurol 2006;63:435-40.
- Lenze EJ, Rogers JC, Martire LM, Mulsant BH, Rollman BL, Dew MA, et al. The association of late-life depression and anxiety with physical disability: a review of the literature and prospectus for future research. Am J Geriatr Psychiatry 2001;9:113-35.
- Lenze EJ, Schulz R, Martire LM, Zdaniuk B, Glass T, Kop WJ, et al. The course of functional decline in older people with persistently elevated depressive symptoms: longitudinal findings from the Cardiovascular Health Study. J Am Geriatr Soc 2005;53:569-75.
- Köhler S, Thomas AJ, Barnett NA, O’Brien JT. The pattern and course of cognitive impairment in late life depression. Psychol Med 2010;40:591-602.
- Bhalla, RK, Butters MA, Mulsant BH, Begley AE, Zmuda MD, Schoderbek B, et al. Persistence of neuropsychologic deficits in the remitted state of late-life depression. Am J Geriatr Psychiatry 2006;14:419-27.
- Panza F, D’Introno A, Colacicco AM, Capurso C, Del Parigi A, Caselli RJ, et al. Depressive symptoms, vascular risk factors, and mild cognitive impairment. The Italian longitudinal study on aging. Dement Geriatr Cogn Disord 2008;25:336-46.
- Lyness TM, Kim J, Tang W, Tu X, Conwell Y, King DA, et al. The clinical significance of subsyndromal depression in older primary care patients. Am J Geriatr Psychiatry 2007;15:214-23.
- Rosenberg PB, Mielke MM, Xue QL, Carlson MC. Depressive symptoms predict incident cognitive impairment in cognitive healthy older women. Am J Geriatr Psychiatry 2010;18:204-11.
- Butters MA, Becker JT, Nebes RD, Zmuda MD, Mulsant BH, Pollock BG, et al. Changes in cognitive functioning following treatment of late-life depression. Am J Psychiatry 2000;157:1949-54.
- Kral VA, Emery OB. Long-term follow-up of depressive pseudodementia of the aged. Can J Psychiatry 1989;34:445-6.
- Herrmann LL, Le Masurier M, Ebmeier KP. White matter hyperintensities in late life depression: a systematic review. J Neurol Neurosurg Psychiatry 2007;79:619-24.
- Schweitzer I, Tuckwell V, Ames D, O’Brien J. Structural neuroimaging studies in late-life depression: a review. World J Biol Psychiatry 2001;2:83-8.
- Panza F, Capurso C, D’Introno A, Colacicco AM, Zenzola A, Menga R, et al. Impact of depressive symptoms on the rate of progression to dementia in patients affected by mild cognitive impairment. The Italian Longitudinal Study on Aging. Int J Geriatr Psychiatry 2008;23:726-34.
- Rozzini L, Chilovi BV, Trabucchi M, Padovani A. Depression is unrelated to conversion to dementia in patients with mild cognitive impairment. Arch Neurol 2005;62:505.