Hong Kong J Psychiatry 2004;14(1):16-22
ORIGINAL ARTICLE
Dr Roger MK Ng, Senior Medical Officer, Kowloon Hospital, 147 Argyle Street, Hong Kong, China.
Address for correspondence: Dr Roger MK Ng, Senior Medical Officer, Kowloon Hospital, 147 Argyle Street, Hong Kong, China.
Submitted: 9 January 2003; Accepted: 22 April 2004
Abstract
Objective: There is a paucity of literature exploring the relationship between early visual processing measures (span of apprehension test and degraded-continuous performance test) and community functioning. The existing literature draws mixed and conflicting conclusions about the significance of early visual processing measures on community functioning.
Patients and Methods: In a group of 66 patients with chronic schizophrenia recruited from a rehabilitation unit in a psychiatric hospital in Hong Kong, baseline measures of positive symptoms, negative symptoms, and early visual processing measures were collected within 2 weeks of recruitment into the study. Day care and night care service data were collected from the hospital database and verified with outpatient case records and information collected from care staff. Results: Principal component analyses of the baseline measures yielded 4 factors corresponding to early visual processing factor, positive symptoms, negative symptoms, and degraded-continuous performance test measures. Early visual processing measures made up of degraded-continuous performance test overall sensitivity and correct rates of span of apprehension test matrix 3, span of apprehension test matrix 12, and span of apprehension test overall correct rate correctly classified 40% of day care services and 62% of hospitalisation status.
Conclusion: The possible mechanism of early visual processing measures in mediating care service requirements is discussed.
Key words: Care services, Schizophrenia
Introduction
There is an increasing surge of interest in the functional consequences of neurocognitive deficits in schizophrenia. There is emerging evidence that neurocognitive measures that are most significantly related to functional outcomes are secondary verbal memory, working memory, executive function, and vigilance.1 Functional outcomes are defined as success in psychosocial skill acquisition, laboratory assessment of instrumental skills and social problem solving ability, and community outcome/daily activities. In a review of 37 studies,1 the meta-analysis concluded that weighted effect sizes of the 4 neurocognitive domains are not negligible (pooled estimate ranged from 0.29 to 0.40).
Among the 4 neurocognitive variables, vigilance has the smallest effect size (0.20), due to the limited number of available studies (n = 17). Of these 17 studies, 9 supported a positive relationship between vigilance and community outcome, while 8 found no relationship. Vigilance refers to the ability to maintain a response to a signal and not respond to noise. Vigilance is typically measured by the continuous performance test (CPT).2 Early visual processing is another neurocognitive domain that has been mentioned in the review to have some positive relationship with social problem solving, but due to the limited number of studies (n = 4), this variable is not powerful enough for subsequent meta- analysis. It is interesting to note that in 3 of the 4 studies included in Green’s meta-analysis, there is a strong positive correlation between early visual processing and social problem solving skills.1 Visual processing deficit in schizophrenia is measured by the span of apprehension test (SPAN), a test of ability to visually scan a display of competing stimuli and identify the visual target stimulus (a ‘T’ or an ‘F’).3 In Bowen et al’s study of chronic stable inpatients with schizophrenia, there was a strong correlation between degraded-CPT and interpersonal performance scales (r = 0.49-0.58, p < 0.01), and strong correlation between degraded-CPT and elemental skills training measures (r = 0.50-0.66, p < 0.01), as well as between SPAN and some elemental skills training measures (listening skills
There are relatively few studies that examine the prognostic value of early visual processing and subsequent level of care. This is important not only because we may predict success but also to realise the full potential of the whole group of patients with chronic illness and to plan services more effectively.10 Response processing difficulties measured by complex reaction time has been found to predict the level of care after 3 years,11 and 6 years.10 In a longitudinal study of patients with acute psychosis, early visual processing (as measured by backward masking) at the time of intake predicted the duration of hospital stay and the level of care at week 6 of the study.12 However, no prospective study of a longer period has ever examined the relationship between early visual processing deficits and degree of independent living in a chronic schizophrenic inpatient population.
Multiple studies have found a weak relationship between positive symptoms and functional outcome.13-17 There is also a moderate and yet uncertain relationship between negative symptoms and functional outcome.17 As the assessment between negative symptoms overlap considerably with social functioning, there is a risk of redundancy of assessment.17 In a 5-year follow-up study of a group of patients with chronic schizophrenia, negative symptoms were significantly worse in those who were resettled in supervised community placement.18 There is evidence to suggest that early percep- tual deficits act as vulnerability factors for development of negative symptoms.19 Furthermore, neurocognitive deficits predated the onset of negative symptoms and schizophrenia in the New York High-Risk Study.20,21 This relationship is also indirectly suggested by the strong relationship between negative symptoms and neurocognitive deficits. In Censit et al’s study, 10 of the 15 significant correlations between clinical improvement and cognitive improvement were with negative symptoms.22
This longitudinal study aimed to test whether early visual processing measures, namely SPAN and degraded-CPT predict discharge from day and night care service require- ments after 1 year. It is also hypothesised that negative symptoms were also correlated with discharge status and work status after 1 year, although to a lesser extent. Positive symptoms are hypothesised to be unrelated to community functioning.
Patients and Methods
Patients
Sixty six consecutive patients admitted to a rehabilitation unit in Hong Kong with a Diagnostic and Statistical Manual of Mental Disorders (DSM IV) diagnosis of schizophrenia were recruited into the study. In addition to the diagnosis, other inclusion criteria included:
- age between 18 and 65 years <
- no history of substance or alcohol abuse in the past year
- absence of brain diseases
- no history of seizure disorder or epilepsy.
The author was responsible for recruiting patients who fulfilled the inclusion and exclusion criteria. The diagnosis of schizophrenia was ascertained after interview with the patients, key nurses, and psychiatrists, and after perusing the case notes.
Procedures and Instruments
Patients were invited to voluntarily participate in the study. After informed consent was obtained from the patients, they were administered the 24-item Brief Psychiatric Rating Scale (BPRS),23 a global symptom scale assessing symptoma- tology on a series of 7-point scales, by 2 research assistants. Prior to making a rating of the BPRS, the 2 research assistants were required to reach a median intra-class correlation coefficient of 0.80 with the author (experienced in using the BPRS) for a series of 10 interviews. Both research assistants were unaware of the purpose of the study. The BPRS-positive score was computed by addition of the scores of unusual thought content, hallucinations, and conceptual disorganisations. The BPRS-negative score was computed by addition of the scores of self-neglect, emotional withdrawal, blunted affect, and motor retardation.
The Rehabilitation Evaluation Hall And Baker (REHAB) scale24 was administered by each patient’s key nurse within 2 weeks of recruitment into the study. REHAB is a global rating scale that has been validated in a local hospital population in Hong Kong.25 The REHAB scale was selected for this hospital-wide survey. This scale has been used for global assessment of social functioning in a chronic psychiatric population and has been shown to have good inter-rater and test-retest reliability.26 The REHAB scale comprises 2 item formats, one on deviant behaviour (DB) and the other on general behaviour (GB). The DB item screens for the presence and the degree of severity of 7 deviant problem areas, namely incontinence, absconding, sexual acts, talking to self, violence, verbal aggression, and self-injury. Each problem can be rated according to the degree of severity (no or mild problem = 0; moderate problem with target behaviour occurring once in a week = 1; severe problem with target behaviour occurring more than once in a week = 2). The GB item can be divided into 5 factors: self-care, community skills, speech disturbance, social activity, and overall rating. Under each factor, there are several factor items. The 5 factors therefore give rise to 16 factor items. For each factor item, a rating ranging from 0 to 7 can be assigned by the nurse rater on a visual analogue scale, using a line with 3 cues under the line, one at the left, one in the middle, and one at the right. In the self-care factor, there are 5 factor items: table manners, washing, dressing, looking after possessions, and prompting. In the factor of community skills, 2 items are assessed: money use and use of public facilities. In the speech disturbance factor, there are 2 items: amount of speech and clarity of speech. In the factor of social activity, there are 6 items: mixing on ward, mixing off ward, spare time, amount of speech, initiation of speech, and level of activity. Finally, the factor of overall rating allows the rater to give a global clinical impression of the patient. All the nurses involved must attain a criterion inter-rater reliability of at least 0.80 (Spearman rank correlation) before they are included for the rating.
The participants were also administered 2 early visual processing measurements, the degraded-CPT and SPAN, within 2 weeks after recruitment into the study. Degraded- CPT and SPAN were administered under conditions specified by the University of California, Los Angeles (UCLA) group2 in terms of computer monitor type, the level of illumination of the computer screen, and the laboratory environment, so as to ensure comparability with the UCLA group. The 2 research assistants, who were not aware of the study hypothesis under investigation, administered the degraded-CPT and SPAN. The author supervised 30% of the sessions at random so as to ensure proper execution of the study procedures. Before the commencement of the degraded-CPT and SPAN, each patient was required to read from the Snellen chart to ensure normal visual acuity (with or without any correction glasses). All participants had to pass this visual acuity test to be recruited. Degraded-CPT is a computer-based programme that assesses visual vigilance during an 8-minute period. Participants were shown a series of single digits at a rate of 1 per second and instructed to press a response button whenever they saw the target number ‘0’. Each digit was shown for a duration of 33 milliseconds. The presentation stimuli were degraded to a standard degree by reversing the black/white background for a random 40% of pixels. Hit rate, false alarm rate, and overall sensitivity score (i.e., the overall index of the ability to discriminate hits from false alarms) were used as dependent measures. The degraded-CPT version was validated in a local sample of people with schizophrenia.27
SPAN is a computer-based measure of early visual processing, visual scanning, and iconic readout, requiring subjects to identify which of the 2 target letters (T or F) appears on the monitor by pressing 1 of the 2 response buttons (marked T or F) on a control pad as quickly as possible.28 All subjects must be able to read the 2 English letters (T or F) before they are administered the SPAN. Administration procedures involved a series of practice trials followed by 64 test trials. The 64 test trials consisted of an equal number of 3- and 12-letter arrays that were counter- balanced in order. Each array was presented for 70 milliseconds and the location of the target letters was randomly distributed across trials. The dependent measures were the correct responses for the 3- and 12-letter arrays. SPAN was validated in this patient population by recruiting a separate sample of 17 patients with DSM-IV criteria for schizophrenia and 17 normal controls, matched in age, sex, and years of education. The correct responses in the 3- and 12-letter arrays were significantly worse in the group with schizophrenia (SPAN matrix 3: 0.976 vs 0.930; SPAN matrix 12: 0.802 vs 0.687; SPAN overall rate: 0.889 vs 0.815; all p < 0.05). There was no gender difference in terms of correct response for both matrix 3 and matrix 12 (22 men and 12 women) [SPAN matrix 3: 0.984 vs 0.972; SPAN matrix 12: 0.836 vs 0.784; SPAN overall rate: 0.909 vs 0.878].
Service use was divided into day and night care, modified locally according to the criteria used by Wykes et al10 and Wykes and Dunn.11 These were classified on a scale of dependence on psychiatric services as follows, with some modifications due to local service variations:
- day care — on the ward, no day care; ward helper; ward occupational therapy; hospital industrial therapy; structured tasks in a day centre; sheltered workshop; supported employment; and open employment or living in own home supported by social security
- night care — acute inpatient psychiatric ward; long stay ward; rehabilitation ward; staffed private hostel; halfway houses; and living in own accommodation.
The work status and the residential status of the participants were traced from the hospital computer database after a period of 1 year. If the participants were still in hospital, their current ward status and work status were confirmed from the hospital database, followed by examination of case records in the ward. If they were discharged to the community, the current residential/work provision was confirmed by perusing the outpatient notes, calling the residential services, or checking the address against the data records of the Medical Social Services. The medication dosage and the types of antipsychotics were obtained by contacting the relevant care service providers.
No neuropsychological data were available to the staff who made the decisions about the appropriate services. Even though the author was the psychiatrist-in-charge of the rehabilitation unit and contributed to the discharge plan of the patients, all care staff decided jointly on the discharge plan. Furthermore, the author only gathered all the data (BPRS, degraded-CPT, and SPAN) by the end of the study. Therefore, the decision of discharge or care programmes was unlikely to be influenced by prior knowledge about neuropsychological performance. The research assistants who conducted the BPRS were not involved in the discharge decision of the subjects. Only the REHAB scores on admission were used for analysis as the key nurses who were involved in the discharge decision would not have biased the rating on initial admission.
Statistics
For categorical variables such as sex and types of antipsychotics, Chi squared tests were used for detection of statistical differences. Upon examination of the scatter diagrams, some continuous data (dosage of antipsychotics, dosage of benzhexol, CPT indices, SPAN indices, BPRS- positive scores) were highly skewed, so non-parametric analyses were adopted for detection of significant differences. For continuous data with normal distribution (duration of illness, age), parametric tests were adopted. Spearman rank correlation was used for all correlational analyses in view of the highly skewed data. Due to the exploratory nature of this study, the p value was set at 0.05 without Bonferroni correction.
Results
Patients’ Characteristics
The study group was made up of 14 women and 52 men. All were ethnic Chinese people. The mean age was 38.30 years (standard deviation [SD], 9.11 years); the mean years of education was 10.2 years (SD, 2.3 years). The mean age of onset of illness was 15.2 years (SD, 7.2 years). The mean duration of hospital stay was 600.0 days (SD, 465.7 days). After an initial screening at admission to the rehabilitation ward, the study diagnosis was screened by the author to confirm the diagnosis according to the DSM-IV criteria. Diagnosis was made using interview data and clinical records. All patients were taking maintenance doses of neuroleptic medication, with mean chlorpromazine equivalents of 566.9 mg (SD, 369.2 mg). The mean dosage of benzhexol was 1.03 mg (SD, 1.99 mg). Forty six patients were given atypical antipsychotics (24 were given clozapine, 12 olanzapine, 5 risperidone, and 2 quetiapine) and 20 were taking typical antipsychotics. There was no significant dif- ference in the age (Mann-Whitney test; p = 0.434), sex (Chi squared test; p = 0.342), chronicity of illness (Mann-Whitney test; p = 0.116), and duration of hospital stay (Mann-Whitney test; p = 0.620) in index admission between those patients taking conventional and atypical antipsychotic agents. Patients taking atypical and conventional antipsychotic agents were therefore pooled in the subsequent analysis.
Characteristics Between Discharged and Hospitalised Patients at 1-year Follow up
Patients were traced 1 year after admission to obtain information on their current residential and work status. Twenty nine patients were discharged to various residential accommodations (private hostel, halfway houses, com- passionate flats). Only 2 patients discharged were briefly re-admitted to hospital for stabilisation but they were discharged back to the original placement at the time of follow up at 1 year. No patients moved out of their discharged community placement during the follow-up period. There were no significant differences between the 2 groups (discharged or continuing hospitalisation) in terms of age (t = -0.943; p = 0.349) or duration of illness (t = -0.264; p = 0.793). There was also no significant difference in the baseline dosage of antipsychotics in terms of chlorpromazine equivalents (z = -6.82; p = 0.495; Mann-Whitney test) and dosage of benzhexol (z = -1.788; p = 0.074; Mann-Whitney test). There were no significant differences between the 2 groups in terms of sex (Chi squared, p = 0.343) and number of patients taking atypical antipsychotics (Chi squared, p = 0.233). One patient in the discharged group and 1 patient in the hospitalised group switched to atypical antipsychotics. Five patients in the discharged group had the antipsychotic dosage increased versus 4 patients in the hospitalised group (Fisher exact test, p = 0.54). There were no correlations between change in dosage of antipsychotic agents and the types of day or night care services required (Spearman rank correlation, p = 0.625). The CPT-hit rate, the degraded stimuli-continuous performance test (DS-CPT) sensitivity, SPAN matrix 3 and SPAN matrix 12 were all worse in the persistently hospitalised groups. The hospitalised group was also more severe for negative symptoms. No difference was detected for positive symptom score and total REHAB score in the 2 groups (Table 1).
Neuropsychological Performance Between Patients Taking Atypical and Typical Antipsychotics
Comparison of patients taking atypical antipsychotics versus those taking conventional antipsychotics found no significant difference in the neuropsychological indicators (CPT-hit rate: 0.535 vs 0.544; DS-CPT overall sensitivity: 0.742 vs 0.754; SPAN matrix 3: 0.879 vs 0.873; SPAN matrix 12: 0.653 vs 0.647; SPAN overall rate: 0.731 vs 0.734), suggesting that these indicators were not associated with the use of a particular type of antipsychotic agent.
Relationship Between Symptom Scores, Early Visual Processing Measures, and Service Use after 1 Year
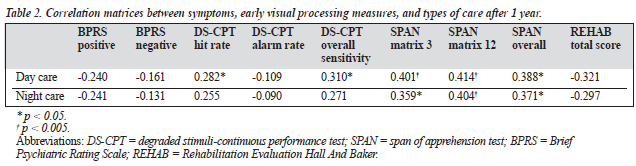
Only neuropsychological indicators correlated with service use. Specifically, CPT-hit rate and DS-CPT overall sensitivity correlated with day care service; SPAN matrix 3, SPAN matrix 12, and SPAN overall rate correlated with day and night care services. The statistical powers all reached a p value of less than 0.05. These correlations remained sig- nificant after partial correlation controlling for negative score of BPRS. Positive and negative scores derived from the BPRS and the behavioural measure of REHAB are not associated with subsequent care service requirements (Table 2).
Predictors of Care Services Required by Early Visual Processing Factor
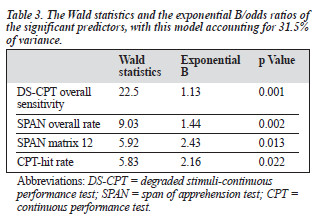
Logistic regression was adopted to consider the predictive value of the various independent factors in predicting the use of community care services after 1 year. The dependent measure was the use of community care/hospital admission and the independent measures included age, sex, duration of hospital stay, type of antipsychotics used, positive and negative scores of the BPRS, and the various neuro- psychological indicators found to be significantly different between hospitalised and discharged patients (CPT-hit rate, DS-CPT overall sensitivity, SPAN matrix 3 correct rate, SPAN matrix 12 correct rate, SPAN overall correct rate). Only CPT-hit rate, DS-CPT overall sensitivity, SPAN matrix 12, and SPAN overall correct rate were significant predictors of use of community services after 1 year (Table 3).
Discussion
The results supported the hypotheses that early visual processing measures, namely SPAN and degraded- CPT, were possible predictors of community outcome of patients with chronic schizophrenia in a chronic inpatient rehabilitation setting in Hong Kong. The positive symptoms were not predictive of community outcome as defined by day and night care service requirements. However, negative symptoms did not enter into the predictor set of the community service requirements. This is perhaps not surprising, as this study employed a very crude measure of negative symptoms (motor retardation, blunted affect, emotional withdrawal, and self-neglect sub-scores to compose a BPRS negative symptom score). Use of a more comprehensive scale for negative symptoms might provide a more convincing answer to this controversial issue.
The success of the early visual processing measure in predicting day service requirements and discharge status can be related to its mediating effect in social skills acquisition.4,6,29 The effect of cognitive deficits on skill learning may be related to serial models of social cognition, in which the first stage of processing is social cue perception, 29 which is an indicator tapping similar neurocognitive constructs as SPAN and degraded-CPT. This hypothesis is further supported by a recent study in which SPAN matrix 12 correct rate alone accounted for 50% of the variance of the facial emotion identification test.30 Perception of emotion may require the ability to extract salient information from briefly presented stimuli, and deficits in early visual processing may lead to a detrimental cascading effect on the perception of emotion/social cognition. Social competence is essential for successful community adaptation.31
The finding of lack of significant association between type of antipsychotic agent taken and residential outcome deserves further investigation as there are multiple studies supporting improvement in cognitive function after switching to atypical antipsychotics,32,33 and the augmentation effect of atypical antispychotics on benefits accrued from rehabilitation training. The lack of association may be related to the relatively short duration of use of atypical antipsychotic agents in this study group (mean period, 8.9 months), as most studies reporting positive effect on cognition usually examined patients taking atypical antipsychotics for more than 1 year. Another possibility of non-association is that early visual processing measures such as SPAN and degraded-CPT are vulnerability markers of schizophrenia that are enduring and resistant to medication effect.1
There are several limitations to this study. The small group was drawn from a rehabilitation unit in a psychiatric hospital so that the patients were mostly chronic schizo- phrenic inpatients with relatively poor psychosocial functioning. The result could therefore not be applied to the recent-onset schizophrenic population. There was no symptom rating at the 1-year follow-up point so whether change in psychotic or negative symptoms contributes to care service requirement cannot be answered.
Acknowledgements
The author would like to thank the 2 research assistants, Mr Paul Kong and Miss Josephine Yau, for their hard work. The author would like to thank the contribution of the ward nurses of the Hospital Pre-discharge Unit of Castle Peak Hospital to this research. The author would also like to thank Professor KH Neuchterlein of UCLA for providing the SPAN and degraded-CPT software for this study.
References
- Green MF. Neurocognitive indicators of vulnerability to schizophrenia. In: Green MF, editor. Schizophrenia from a neurocognitive perspective. Boston: Allyn & Bacon; 2000:61-82.
- Nuechterlein KH. Signal detection in vigilance tasks and behavioral attributes among offspring of schizophrenic mothers among hyperactive children. J Abnormal Psychol 1983;92:4-28.
- Asarnow RF, Granholm E, Sherman T. Span of apprehension in schizophrenia. In: Steinhauer SH, Gruzelier JH, Zubin J, editors. Handbook of schizophrenia. Vol 5. Neuropsychology, psycho- physiology, and information processing. Amsterdam: Elsevier 1991;335-370.
- Bowen L, Wallace CJ, Glynn SM, Nuechterlein KH, Lutzer JR, Kuehnel TG. Schizophrenic individuals’ cognitive functioning and performance in interpersonal intereactions and skills training procedures. J Psych Res 1994;28:289-301.
- Green MF, Kern RS, Braff DL, Mintz J. Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the right stuff? Schizophr Bull 2000;26:119-136.
- Kern RS, Green MF, Satz P. Neuropsychological predictors of skills training for chronic schizophrenic patients. Psychiatry Res 1992;43: 223-230.
- Corrigan PW, Green MF, Toomey R. Cognitive correlates to social cue perception in schizophrenia. Psychiatry Res 1994;53:141-151.
- Neuchterlein KH, Dawson ME, Green MF. Informational processing abnormalities as neuropsychological indicators for schizophrenia. Acta Psychiatrica Scand 1994;90:71-79.
- Addington J, Addington D. Neurocognitive and social functioning in schizophrenia. Schizophr Bull 1999;32:173-182.
- Wykes T, Sturt E, Katz R. The prediction of rehabilitation success after three years: the use of social, symptomatic and cognitive variables. Br J Psychiatry 1992:157:865-870.
- Wykes T, Dunn G. Cognitive deficits and the prediction of rehabilitation success in a chronic psychiatric group. Psychol Med 1992;22:389-398.
- Spaulding WD, Fleming SK, Reed D, Sullivan M, Storzbach D, Lam M. Cognitive functioning in schizophrenia: implications for rehabili- tation. Schizophr Bull 1999;25:275-290.
- Bell MD, Lysaker PH. Psychiatric symptoms and work performance among persons with severe mental illnesses. Psychiatric Serv 1995; 46:508-510.
- Dickerson F, Boronow JJ, Ringel N, Parente F. Neurocognitive deficits and social functioning in outpatients with schizophrenia. Schizophr Res 1996;48:75-83.
- Brekke JS, Raine A, Ansel M, Lencz T. Neuropsychological correlates and psychophysiological correlates of psychosocial functioning in schizophrenia. Schizophr Bull 1997;23:19-28.
- Velligan DI, Mahurin RK, Diamond PL, Hazelton BC, Eckert SL, Miller AL. The functional significance of symptomatology and cognitive functions in schizophrenia. Schizophr Res 1997;25:21-31.
- Green MF, Nuechterlein KH. Should schizophrenia be treated as a neurocognitive disorder? Schizophr Bull 1999;25:309-318.
- Ng RMK, Leung SP. The 5-year outcome study of a group of chronic psychotic patients discharged into the community. HK J Psychiatry 2000;10:27-36.
- Nuechterlein KH, Edell WS, Norris M, Dawson ME. Attentional vulnerability indicators, thought disorder and negative symptoms. Schizophr Bull 1986:12:408-426.
- Cornblatt B, Lenzenweger MF, Dworkin R, Erlenmeyer-Kimling L. Childhood attentional dysfunction predicts social deficits in unaffected adults at risk for schizophrenia. B J Psychiatry 1992;161 (Suppl 18): 59-64.
- Erlenmeyer-Kimling L, Rock D, Roberts S, Janal M, Kestenbaum C, Cornblatt B. Attention, memory, and motor skills as childhood predictors of schizophrenia-related psychoses: the New York High-Risk Project. Am J Psychiatry 2000;157:1416-1422.
- Censits DM, Ragland JD, Gur RC, Gur RE. Neuropsychological evidence supporting a neurodevelopmental model of schizophrenia: a longitudinal study. Schizophr Res 1997;24:289-298.
- Lukoff D, Liberman RP, Nuechterlein KH. Symptom monitoring in the rehabilitation of schizophrenic patients. Schizophr Bull 1986; 12:578-602.
- Baker B, Hall R. Part 2: technical information: validity. In: Baker B, Hall R, editors. User manual for Rehabilitation Evaluation Hall And Baker. Aberdeen: VINE: 1988;119-121.
- Ng RMK, Leung SP. REHAB scale survey in Castle Peak Hospital in Hong Kong. Hong Kong J Psychiatry 2002;12:14-22.
- Carson J, Coupar A, Gill J, Titman P. The inter-rater reliability of Hall and Baker’s REHAB scale: a cross-validation study. Br J Clin Psychol 1988;27:277-278.
- Ng RMK. Degraded continuous performance test and psychosis- proneness among undergraduates in Hong Kong. Hong Kong J Psychiatry 2002;12:6-13.
- Asarnow RF, Neuchterlein KH. Manual and computer program for the Forced-Choice, Partial Report Span of Apprehension Test (Version 3.4). Los Angeles: Universtiy of California Los Angeles; 1996.
- 29. Corrigan PW, Schade ML, Liberman RP. Social skills training. In: Liberman RP, editor. Handbook of psychiatric rehabilitation. Boston: Allyn & Bacon; 1992:95-126.
- Kee KS, Kern RS, Green MF. Perception of emotion and neurocognitive functioning in schizophrenia: what’s the link? Psychiatry Res 1998;81:57-65.
- Penn DL, Corrigan PW, Bentall RP, Racenstein JM, Newman L. Social cognition in schizophrenia. Psychological Bull 1997;121:114-132.
- Green MF, Marshall BDJ, Wirshing WC, et al. Does risperidone improve verbal memory in treatment resistant schizophrenia? Am J Psychiatry 1999;154:799-804.
- Purdon SE, Jones BD, Stip E, et al Neuropsychological change in early phase schizophrenia during 12 months of treatment with olanzapine, risperidone, or haloperidol. Arch Gen Psychiatry 2000;57:249-258.