Hong Kong J Psychiatry 2002;12(4):2-5
ORIGINAL ARTICLE
S Xiao, P Yao, X Li, M Zhang
This study was supported by the research grant (98-1-320) of China Health Ministry.
Dr S Xiao, Shanghai Mental Health Center, Shanghai, China.
Dr P Yao, Shanghai Mental Health Center, Shanghai, China.
Dr X Li, Shanghai Mental Health Center, Shanghai, China.
Dr M Zhang, Shanghai Mental Health Center, Shanghai, China.
Address for correspondence: Dr S Xiao, 600 Wan Ping Nan Road, Shanghai Mental Health Center, Shanghai 20030, China.
Submitted: 21 March 2001 ; Accepted: 03 September 2001
Abstract
Objective: To identify markers of early cognitive changes in Alzheimer’s disease using a battery of neuropsychological tests.
Methods: The study was cross-sectional with a case-control design, and evaluated 3 groups of subjects: patients with mild and moderate Alzheimer’s disease, those with mild cognitive impairment, and normal controls. The World Health Organization Battery of Cognitive Assessment Instruments for elderly people was used to assess the cognitive function of the study patients. Results: Twenty six individuals with Alzheimer’s disease, 27 with mild cognitive impairment and 83 controls were recruited. Mean total scores of the Mini-Mental Status Examination were 17.6 (SD, 2.0), 24.2 (SD, 2.1), and 28.7 (SD, 1.1), respectively. The results of the World Health Organization Battery of Cognitive Assessment Instruments tests demonstrated significant differences among the 3 groups for all subtests. Similarly, there were significant differences between the Alzheimer’s disease group and either the control group or the mild cognitive impairment group for nearly all subtests. However, significant differences between those with mild cognitive impairment and control subjects were found only for some subtests of the battery; these included auditory verbal learning, delayed recall, sorting, verbal fluency, mini-token, trail making, visual reasoning, and spatial construction. The group with mild cognitive impairment performed significantly worse than controls for these tests.
Conclusion: The findings of this study suggest that auditory verbal learning, delayed recall, sorting, verbal fluency, mini-token, trail making, visual reasoning and spatial construction tests are the more sensitive subtests for early detection of Alzheimer’s disease.
Key words: Alzheimer disease, Dementia, presenile, Neuropsychological tests
Introduction
Alzheimer’s disease (AD) is a major illness of the modern era. Most studies have found that nearly 5% of people aged 65 years or older have dementia, of which AD is the most common cause. In China, community population surveys indicate that up to 3% to 5% of elderly community residents aged 60 years and older have AD or closely related dementias of late life.1 Since 130 million people in China are older than 60 years, it is estimated that approximately 3 to 4 million people in China suffer from AD.
While its aetiology is still unknown, the pathological manifestations of AD seen at autopsy are well documented: the characteristic brain atrophy, amyloid plaques, and neurofibrillary tangles. Atrophy is usual with advancing age but can be very severe in AD. Cerebral convolutions are narrowed and sulci are widened. Third and lateral ventricles are asymmetrically enlarged. The atrophic process involves the frontal, temporal and parietal lobes. A widespread loss of nerve cells is observed microscopically. Residual neurones probably lose volume, dendrites, and ribonucleoprotein. Proliferation of astrocytes is a sign of compensation or repair.
The diagnosis of AD at its later stages usually does not pose much difficulty and the accuracy of making the clinical diagnosis of AD is reasonably high. However, difficulties remain in early detection of AD at a presymptomatic phase, particularly in differentiating early AD from the cognitive change found in normal ageing. Early diagnosis is very important because the deterioration of AD may only be postponed or arrested by treatment in its early stage.2
Since decline in cognitive function (especially in memory) is usually the earliest manifestation of AD, we suggest that neuropsychological tests may give valuable evi- dence for early detection of AD. Mild cognitive impairment (MCI) is thought to be a cognitive deficit state between AD and normal senility. Elderly subjects with MCI develop dementia at a rate of 8% to 15% per year, compared with only about 1% for those with normal cognitive function.3
The present paper reports the efforts to identify neuro- psychological markers for the purpose of differentiating individuals with MCI from those with Alzheimer’s disease and normal elderly people.
Methods
Subjects
Subjects included 26 patients with mild to moderate Alzheimer’s disease (AD group), 27 elderly people with mild cognitive impairment (MCI group) and 83 normal controls (NC group). Inclusion criteria for the AD group were as follows:
- a definite diagnosis of AD according to DSM-IV4 and the National Institute of Neurological and Communicative Disorders and Stroke (NINCDS)5 criteria
- age 55 to 80 years
- Mini-Mental State Examination (MMSE)6 score between 15 and 25
- Hachinski Ischaemic Scale (HIS)7 score less than 4
- Global Deterioration Scale (GDS)8 rating of 3 to 4. Inclusion criteria for the MCI group were as follows:
- age 55 to 80 years
- a history of memory decline lasting more than 3 months but not fully meeting DSM-IV criteria for dementia
- MMSE score less than 26
- Activity of Daily Living Scale (ADL-21 items)9 score less than 26
- GDS rating of 2 to 3
- Absence of specific causes for memory decline or cogni- tive damage.
The NC group was aged 50 to 80 years and did not meet the criteria for the AD and MCI groups. All patients with AD were recruited from inpatients or outpatients of Shanghai Mental Health Center. Subjects in the MCI and NC groups were drawn from memory clinics or the community.
Measures
The measurement instruments included: the World Health Organization Neuropsychological Battery of Cognitive Assessment Instruments (WHO-BCAI) for the elderly,10 composed of verbal learning tests, trail making tests, sorting and concentration tests, language tests, psychomotor tests, visual gnostic function tests and spatial construction tests; the MMSE; ADL; GDS; and HIS.
Data Analysis
Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) version 6.1 software. Continuous demographic variables were examined using the t-test and categorical demographic variables were analysed using the Chi squared test or Fisher exact test. Group differences were tested using 1-way analysis of variance (ANOVA) for comparison among 3 groups and Duncan’s test for comparison between 2 groups.
Results
Sample Characteristics
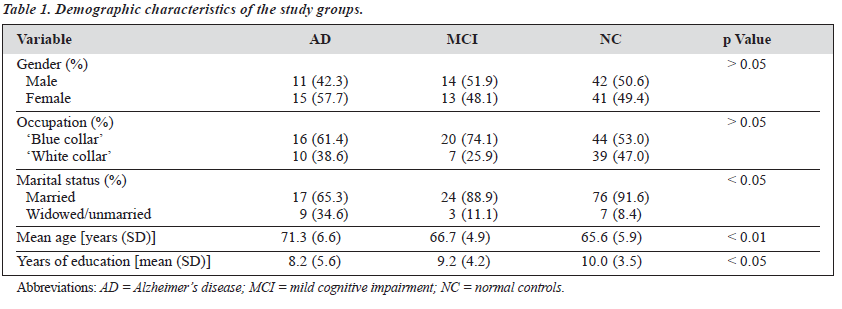
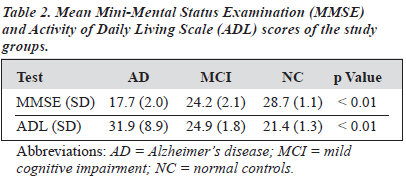
Demographic data and mean MMSE and ADL scores for the 3 groups are summarised in Tables 1 and 2. In total, 26 patients with AD, 27 subjects with MCI and 83 NC were recruited. The mean ages for the AD, MCI and NC group were 71.3 (SD, 6.5), 66.7 (SD, 4.9), and 65.6 (SD, 5.9) years, respectively. There was no significant difference among the 3 groups in gender and occupation, although significant differences were found in marital status, age, and education level. The patients with AD were relatively older and less educated. Mean scores of MMSE were 17.6 (SD, 2.0) for the AD group, 24.2 (SD, 2.1) for the MCI group, and 28.7 (SD, 1.1) for the NC group. ADL assessment scores for the AD, MCI, and NC groups were 31.9 (SD, 8.9), 24.9 (SD, 1.8), and 21.4 (SD, 1.3), respectively. Comparisons of both MMSE and ADL scores revealed significant dif- ferences among the 3 groups. Differences for MMSE scores were significant between any 2 groups, while no significant difference was observed between the MCI and NC groups for ADL scores.
World Health Organization Neuropsychological Battery of Cognitive Assessment Instruments Scores
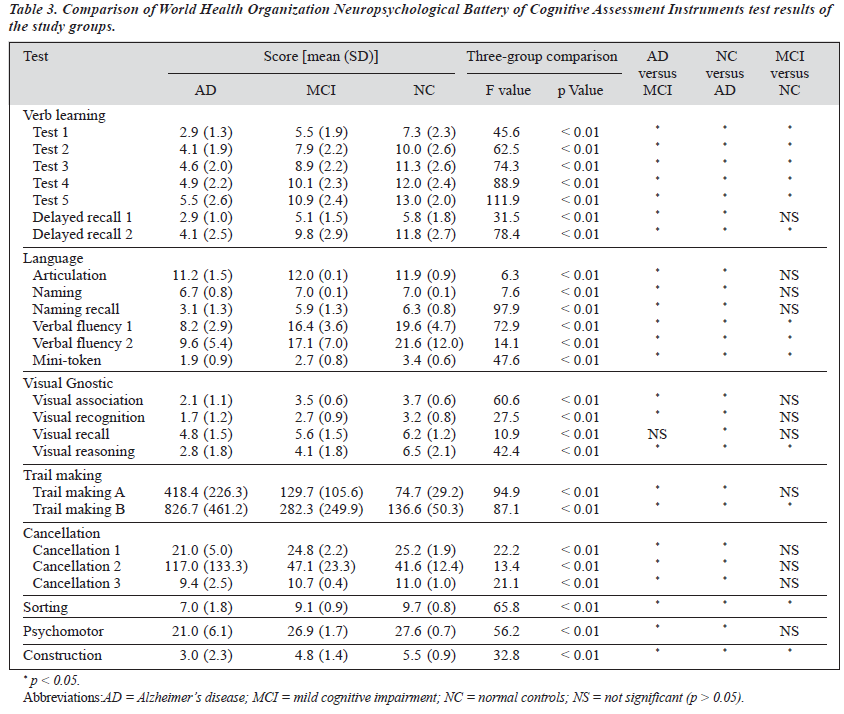
The results of 1-way ANOVA and Duncan’s test with the mean scores of WHO-BCAI for the study groups are depicted in Table 3. Significant differences among the 3 groups in all 25 subtests of the WHO-BCAI were found. AD patients had the lowest scores, and the MCI group scored worse than the NC group. Between-group comparisons suggested that patients with Alzheimer’s disease performed signi- ficantly worse than both the MCI and NC groups in nearly all subtests except visual recognition. However, only 13 subtests showed significant differences between the MCI and NC groups, including verbal learning, verbal fluency, mini-token, visual reasoning, trail making, sorting and spatial construction tests.
Discussion
Mild cognitive impairment (MCI) in elderly people has long been a controversial category in psychiatry. In 1958, Kral proposed the term “benign senile forgetfulness” to denote forgetting details of events and impairment of long-term memory.11 Although initially benign senile forgetfulness was believed to be non-progressive and attributable to normal ageing, it is known that some individuals with benign senile forgetfulness ultimately progress to dementia.11 In the 1980s, the term “age-associated memory impairment” (AAMI) and its research criteria were introduced by an American working group. AAMI defines impairment compared with healthy adult norms, but not with age-matched controls. According to this definition, 16% of young adults will meet the AAMI criteria when they are old without any decline of memory.12,13 Similar terms used to denote cognitive impairment associ- ated with ageing include age-related cognitive decline (DSM-IV) and mild cognitive disorder (ICD-10), but the criteria do not exclude specific causes of the disorder.4,13
In our study, the mild neurocognitive impairment criteria of DSM-IV were adapted by excluding specific causes that can lead to cognitive impairment of elderly people. For example, measures were taken to exclude subjects with evidence of stroke, other neurological disorders, and those who had clinically significant or active renal, hepatic, endocrine, and cardiovascular diseases. In addition, the duration criterion was extended to 3 months. The aim of all of these measures was to increase the homogeneity of the study sample, thus increasing the likelihood that recruited MCI subjects had presymptomatic AD or MCI of Alzheimer type.
The results of the present study demonstrate that the AD group performed worse than the MCI group and the MCI group worse than the NC group for the WHO-BCAI tests. While patients with AD performed significantly worse than both MCI and NC groups in nearly all subtests, significant differences between the MCI and NC groups were found only in some subtests. The MCI group performed signifi- cantly worse than the NC group on verbal learning, verbal fluency, mini-token, visual reasoning, trail making, sorting, and spatial construction tests. There were no differences between these latter groups for tests of pronunciation, naming, attention, motion, and most visual functions. The findings of our study suggest that cognitive impairment of MCI is selective, not pervasive.
Verbal learning, verbal fluency, mini-token, visual reasoning, trail making, sorting, and spatial construction tests are correlated with higher neuropsychological functions such as memory, abstract thinking, planning, reasoning, and executive function. The hippocampus, frontal, and tempo- parietal cortices are associated with such higher neuro- psychological functions.13 The results of our study seem consistent with those of pathological and brain imaging studies which have demonstrated that the hippocampus, frontal cortex, and tempo-parietal cortices are more vulnerable to damage than the primary sensory, motor, and visual cortices in the early stage of AD.15,16
In addition, our results showed poor trail making and spatial construction abilities in subjects with MCI, findings consistent with other reports of abnormal executive function in the early stage of AD.17,18 Therefore, we conclude that verbal learning, verbal fluency, mini-token, visual reasoning, trail making, sorting, and spatial construction tests are more sensitive than other WHO-BCAI subtests for early detection of AD.
There are several limitations to this study. First, the study sample is small. Second, the age and education of the 3 groups were not strictly comparable at inclusion. Third, although we have tried to increase the homogeneity of the study sample to increase the likelihood that the recruited MCI patients actually had pre-syndrome AD, the proportion of MCI patients that progress to AD is still unknown, and cannot be evaluated in this cross-sectional study. Therefore, the results of our study need to be replicated and confirmed using longitudinal study design.
References
- Zhang MY, Katzman R, Jin H, et al. The prevalence of dementia and Alzheimer disease in Shanghai, China. Ann Neurol 1990;27: 428-437.
- Duncan BA, Siegal AP. Early diagnosis and management of Alzheimer’s disease. J Clin Psychiatry 1998;59(Suppl 9):15-21.
- Ritchie K. Neuropsychological assessment in Alzheimer’s disease: cur- rent status and future directions. Int Psychogeriatr 1997;1(Suppl 1): 95-104.
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington: American Psychiatric Association; 1994:706-708.
- McKhann G, Drachman D, Folstein M, et al. Report of the NINCDS- ADRDA work group under the auspices of the Department of Health and Human Services Task Force on Alzheimer Disease. Neurology 1984;34:939-944.
- Folstein MF, Folstein SE, McHugh PR, et al. “Mini mental state” examination: a practical method for grating the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189-198.
- Hachinski VC, Iliff LD, Zilhka E. et al. Cerebral blood flood flow in dementia. Arch Neurol 1975;32:632-637.
- Reisberg B, Ferris SH, De Leon MJ, et al. An instrument for the assessment of primary degenerative dementia (PDD). Am J Psychiatry 1982;139:1136-1139.
- Katz S, Ford AB, Moskowitz RW, et al. Studies of illness in aged: the index of ADL, a standardized measure of biological and psychosocial function. JAMA 1963;185:94-106.
- World Health Organization. World Health Organization-battery of cognitive assessment Instrument for elderly. Geneva: World Health Organization, 1996.
- Kral VA. Senescent forgetfulness: benign and malignant. Can Med Assoc J 1962;86:257-260.
- Coffey CE, Cummings JL. Textbook of geriatric neuropsychiatry. 1st ed. Washington: American Psychiatric Association Press Inc.; 1994: 145-158.
- Hanninen T, Koivisto K, Reinikainen KJ, et al. Prevalence of ageing- associated cognitive decline in an elderly population. Age Ageing 1996;25:201-205.
- Sadavoy J, Lazarrus LW, Jarvik LF, et al. Comprehensive review of geriatric psychiatry. 2nd ed. Washington: American Psychiatric Association Press Inc.; 1996:325-350.
- Jacoby R, Oppenheimer C. Psychiatry in the elderly. 2nd ed. Oxford: Oxford Medical Publications; 1997:37-62.
- Parnetti L, Lowenthal DT, Presciutti O, et al. 1H-MRS, MRI-based hippocampal volumetry, and 99Tc-HMPAO-SPECT in normal aging, age-associated memory impairment, and probable Alzheimer’s disease. J Am Geriatr Soc 1996;44:133-138.
- Grober E, Kawas C. Learning and retention in preclinical and early Alzheimer’s disease. Psychol Aging 1997;12:183-188.
- Kluger A. Gianutsos JG, Golomb J, et al. Motor/psychomotor dys- function in normal aging, mild cognitive decline, and early Alzheimer’s disease: diagnostic and differential diagnostic features. Int Psychogeriatr 1997;1(Suppl 1):307-316.